1,2-Diaminocyclohexane
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Cyclohexane-1,2-diamine | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.010.707 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 2735 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H14N2 |
| Molar mass | 114.192 g·mol−1 |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H302, H302+H312+H332, H312, H314, H317, H332, H335 |
Precautionary statements
|
P260, P264, P264+P265, P270, P271, P272, P280, P301+P317, P301+P330+P331, P302+P352, P302+P361+P354, P304+P340, P305+P354+P338, P316, P317, P319, P321, P330, P333+P317, P362+P364, P363, P403+P233, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
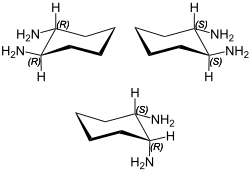
1,2-Diaminocyclohexane (DACH) is an organic compound with the formula (CH2)4(CHNH2)2. It is a mixture of three stereoisomers: cis-1,2-diaminocyclohexane and both enantiomers of trans-1,2-diaminocyclohexane. The mixture is a colorless, corrosive liquid, although older samples can appear yellow. It is often called DCH-99 and also DACH.
Manufacture
The product is available commercially, manufactured by the hydrogenation of o-phenylenediamine. The two trans enantiomers can be resolved by conversion to diastereomeric salts of various chiral acids.[2]
Uses
The product is an epoxy curing agent for use in Coatings, Adhesives, Sealants and Elastomers - CASE.[3] It is particularly useful in epoxy flooring.[4] It may also be reacted with diethyl maleate utilizing the Michael reaction to produce a polyaspartic compound of CAS number 481040-92-0.[5] It may also be used in lubricants.[6] The product is also advertised as being useful as a chelating agent in a variety of applications including oil production.[7] It also is used in downfield oil and gas wells where there is an acidic stream to prevent corrosion to the bore piles.[8]
See also
- Hexamethylenediamine
- Isophorone diamine
- 1,3-BAC
- 2,3-Butanediamine, a vicinal diamine that also exists as three stereoisomers
References
- ^ "1,2-Diaminocyclohexane". pubchem.ncbi.nlm.nih.gov.
- ^ Kouklovsky, Cyrille; Langlois, Yves; Aguilar, Enrique; Fernández-García, Jesús M.; Sikervar, Vikas (2014). "(1S,2S)-1,2-Diaminocyclohexane". Encyclopedia of Reagents for Organic Synthesis. pp. 1–23. doi:10.1002/047084289x.rn00145.pub3. ISBN 978-0-470-84289-8.
- ^ "Dytek DCH-99 by INVISTA - Paint & Coatings". www.ulprospector.com. Retrieved 2020-04-13.
- ^ "A New Epoxy Curing Agent with Long Pot Life and Fast Cure". www.pcimag.com. Retrieved 2021-05-18.
- ^ "Dytek DCH-99 | 1,2-Diaminocyclohexane". Dytek. Retrieved 2020-04-13.
- ^ "US Patent for Non-aromatic based antioxidants for lubricants Patent (Patent # 9,273,266 issued March 1, 2016) - Justia Patents Search". patents.justia.com. Retrieved 2020-04-13.
- ^ "Technical Data Sheet Dytek DCH 99" (PDF). Archived (PDF) from the original on 2020-08-19.
- ^ Materials, Ascend. "FlexaTram-DAM". Ascend Performance Materials. Retrieved 2020-05-21.