2-Pyridylethylamine
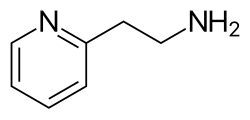 Kekulé, skeletal formula of 2-pyridylethylamine Kekulé, skeletal formula of 2-pyridylethylamine
|
| Names
|
Preferred IUPAC name
2-(Pyridin-2-yl)ethan-1-amine |
| Identifiers
|
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
111208
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| ECHA InfoCard
|
100.018.450
|
| EC Number
|
|
IUPHAR/BPS
|
|
| MeSH
|
2-(2-Aminoethyl)pyridine
|
|
|
|
| UNII
|
|
| UN number
|
2735
|
|
|
|
InChI=1S/C7H10N2/c8-5-4-7-3-1-2-6-9-7/h1-3,6H,4-5,8H2  Y YKey: XPQIPUZPSLAZDV-UHFFFAOYSA-N  Y YInChI=1/C7H10N2/c8-5-4-7-3-1-2-6-9-7/h1-3,6H,4-5,8H2 Key: XPQIPUZPSLAZDV-UHFFFAOYAY
|
NCCc1ccccn1 NCCC1=CC=CC=N1
|
| Properties
|
Chemical formula
|
C7H10N2
|
| Molar mass
|
122.171 g·mol−1
|
| Density
|
1.021 g cm−3
|
| Boiling point
|
93 °C; 199 °F; 366 K at 1.6 kPa
|
| log P
|
−0.11
|
Refractive index (nD)
|
1.536
|
| Hazards
|
| GHS labelling:
|
Pictograms
|

|
Signal word
|
Warning
|
Hazard statements
|
H315, H319, H335
|
Precautionary statements
|
P261, P305+P351+P338
|
| NFPA 704 (fire diamond)
|
|
| Flash point
|
100 °C (212 °F; 373 K)
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
|
2-Pyridylethylamine is a histamine agonist which is selective for the H1 subtype.[1]
References
Histamine receptor modulators |
|---|
| H1 | | Agonists | |
|---|
| Antagonists |
- Others: Atypical antipsychotics (e.g., aripiprazole, asenapine, brexpiprazole, brilaroxazine, clozapine, iloperidone, olanzapine, paliperidone, quetiapine, risperidone, ziprasidone, zotepine)
- Phenylpiperazine antidepressants (e.g., hydroxynefazodone, nefazodone, trazodone, triazoledione)
- Tetracyclic antidepressants (e.g., amoxapine, loxapine, maprotiline, mianserin, mirtazapine, oxaprotiline)
- Tricyclic antidepressants (e.g., amitriptyline, butriptyline, clomipramine, desipramine, dosulepin (dothiepin), doxepin, imipramine, iprindole, lofepramine, nortriptyline, protriptyline, trimipramine)
- Typical antipsychotics (e.g., chlorpromazine, flupenthixol, fluphenazine, loxapine, perphenazine, prochlorperazine, thioridazine, thiothixene)
- Unknown/unsorted: Azanator
- Belarizine
- Elbanizine
- Flotrenizine
- GSK1004723
- Napactadine
- Tagorizine
- Trelnarizine
- Trenizine
|
|---|
|
|---|
| H2 | | Agonists | |
|---|
| Antagonists |
- Bisfentidine
- Burimamide
- Cimetidine
- Dalcotidine
- Donetidine
- Ebrotidine
- Etintidine
- Famotidine
- Isolamtidine
- Lafutidine
- Lamtidine
- Lavoltidine (loxtidine)
- Lupitidine
- Metiamide
- Mifentidine
- Niperotidine
- Nizatidine
- Osutidine
- Oxmetidine
- Pibutidine
- Quisultazine (quisultidine)
- Ramixotidine
- Ranitidine
- Roxatidine
- Sufotidine
- Tiotidine
- Tuvatidine
- Venritidine
- Xaltidine
- Zolantidine
|
|---|
|
|---|
| H3 | | Agonists |
- α-Methylhistamine
- BP 2.94
- Cipralisant (GT-2331)
- GT-2203 (VUF-5296)
- Histamine
- Imetit
- Immepip
- Immethridine (BP-1-5375)
- L-Histidine
- Methimepip
- Proxyfan
- SCH-50971
- VUF-5297
|
|---|
| Antagonists |
- A-349821
- A-423579
- ABT-239
- ABT-652
- AZD5213
- Bavisant
- Betahistine
- Burimamide
- Ciproxifan
- Clobenpropit
- Conessine
- Enerisant
- GSK-189254
- Impentamine
- Iodophenpropit
- Irdabisant
- JNJ-5207852
- NNC 38-1049
- PF-03654746
- Pitolisant
- SCH-79687
- Thioperamide
- VUF-5681
|
|---|
|
|---|
| H4 | | Agonists | |
|---|
| Antagonists |
- JNJ-7777120
- Mianserin
- Seliforant
- Thioperamide
- Toreforant
- VUF-6002
|
|---|
|
|---|
- See also
- Receptor/signaling modulators
- Monoamine metabolism modulators
- Monoamine reuptake inhibitors
|


