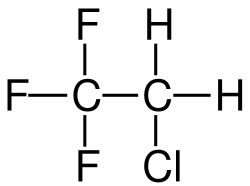
2-Chloro-1,1,1-trifluoroethane
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Chloro-1,1,1-trifluoroethane | |
Other names
| |
| Identifiers | |
CAS Number
|
|
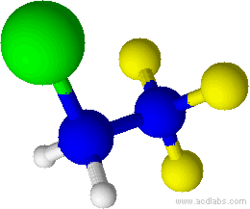
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.000.830 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1983 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C2H2ClF3 |
| Molar mass | 118.48 g/mol |
| Appearance | Gas |
| Density | 1.389 g/mol (liquid at 0 °C) |
| Melting point | −105.3 °C (−157.5 °F; 167.8 K) |
| Boiling point | 6.1 °C (43.0 °F; 279.2 K) |
Solubility in water
|
0.89 g/l |
Refractive index (nD)
|
1.3092 (liquid at 0 °C) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
2-Chloro-1,1,1-trifluoroethane, also known as 1,1,1-trifluoro-2-chloroethane or Freon 133a, is an alkyl halide belonging to the category of hydrochlorofluorocarbons, having chemical formula F3C-CH2-Cl. Under standard conditions, it appears as a colorless gas, partially soluble in water. It is used as a refrigerant, as a solvent and as a reagent in organic synthesis.
It can be made by the reaction of trichloroethylene and hydrogen fluoride.
Gallery
-
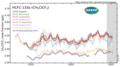 HCFC-133a measured by the Advanced Global Atmospheric Gases Experiment (AGAGE) in the lower atmosphere (troposphere) at stations around the world. Abundances are given as pollution free monthly mean mole fractions in parts-per-trillion.
HCFC-133a measured by the Advanced Global Atmospheric Gases Experiment (AGAGE) in the lower atmosphere (troposphere) at stations around the world. Abundances are given as pollution free monthly mean mole fractions in parts-per-trillion.
References
- ^ "2-Chloro-1,1,1-trifluoroethane | H2ClC-CF3 - PubChem". pubchem.ncbi.nlm.nih.gov.