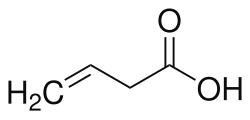
But-3-enoic acid
 | |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.009.902 |
| EC Number |
|
Gmelin Reference
|
362641 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C4H6O2 |
| Molar mass | 86.090 g·mol−1 |
| Appearance | Clear light yellow liquid |
| Density | 1.013 g/mL |
| Melting point | −39 °C (−38 °F; 234 K) |
| Boiling point | 163 °C (325 °F; 436 K) |
Solubility in water
|
Well-soluble |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H314, H317, H335, H341, H351, H411 |
Precautionary statements
|
P202, P273, P280, P303, P304, P305, P310, P338, P340, P351, P353, P361 |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
But-3-enoic acid (or 3-butenoic acid) is a short-chain unsaturated carboxylic acid, with the linear formula CH2=CHCH2CO2H.[2]
Synthesis
The acid is obtained via a reaction of allyl cyanide and concentrated hydrochloric acid. The mixture is gently heated with occasional shaking. The reaction produces a voluminous precipitate of ammonium chloride. Then water is introduced, and the upper acid layer is separated.[3]
Other methods are also known.[4][5]
Properties
The acid forms a clear light yellow liquid. Causes skin burns.[6]
The compound is well-soluble in water, ethanol, and diethyl ether.
Uses
The acid has been employed in preparing specialized coatings and complex organic compounds, including those involving Diels-Alder reactions.[1]
References
- ^ a b "134716 Sigma-Aldrich 3-Butenoic acid". Sigma Aldrich. Retrieved 22 January 2026.
- ^ "3-Butenoic acid". NIST. Retrieved 22 January 2026.
- ^ "Vinylacetic Acid". Organic Syntheses. 24: 96. 1944. doi:10.15227/orgsyn.024.0096. Retrieved 22 January 2026.
- ^ Journal of the Chemical Society. Chemical Society (Great Britain). 1899. p. 736. Retrieved 22 January 2026.
- ^ Journal of the Chemical Society: Abstracts of papers. 1900. p. 8. Retrieved 22 January 2026.
- ^ "Vinylacetic acid - Hazardous Agents | Haz-Map". haz-map.com. Retrieved 22 January 2026.