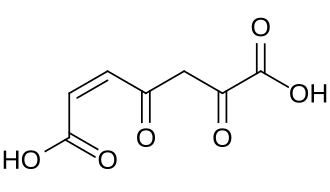
3-Maleylpyruvic acid
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(2Z)-4,6-Dioxohept-2-enedioic acid | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
1725756 |
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C7H6O6 |
| Molar mass | 186.119 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
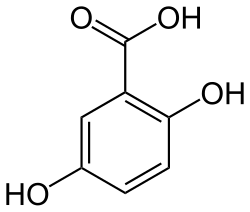
3-Maleylpyruvic acid, or 3-maleylpyruvate, is a dicarboxylic acid formed by the oxidative ring opening of gentisic acid by gentisate 1,2-dioxygenase during the metabolism of tyrosine.[1] It is converted into 3-fumarylpyruvate by maleylpyruvate isomerase.[2]
Biosynthesis
In some organisms, the amino acid tyrosine is metabolised to gentisic acid. In the presence of the enzyme gentisate 1,2-dioxygenase and oxygen, this compound undergoes a ring-opening reaction to give 3-maleylpyruvic acid:[1]
This reaction has been used to detect gentisic acid with a whole-cell biosensor developed from a 3-maleylpyruvic acid-inducible gene expression system and the gentisate 1,2-dioxygenase gene from chemolithoautotrophic bacterium Cupriavidus necator.[3]
Metabolism
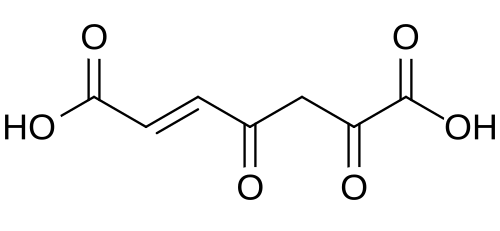
The enzyme maleylpyruvate isomerase converts 3-maleylpyruvic acid to its geometric isomer, 3-fumarylpyruvic acid.[4][2]

References
- ^ a b Sugiyama, Shin-ich; Yano, Keiji; Komagata, Kazuo; Arima, Kei (1960). "Metabolism of Aromatic Compounds by Microbes". Bulletin of the Agricultural Chemical Society of Japan. 24 (3): 243–248. doi:10.1271/bbb1924.24.243.
- ^ a b Lack L (November 1961). "Enzymic cis-trans isomerization of maleylpyruvic acid". The Journal of Biological Chemistry. 236 (11): 2835–40. doi:10.1016/S0021-9258(19)76386-8. PMID 14461395.
- ^ Kutraite, I; Augustiniene, E; Malys,N (2024). "Maleylpyruvic Acid-Inducible Gene Expression System and Its Application for the Development of Gentisic Acid Biosensors". Analytical Chemistry. 96 (47): 18727–35. doi:10.1021/acs.analchem.4c03906. PMC 11603403. PMID 39548649.
- ^ Enzyme 5.2.1.4 at KEGG Pathway Database.