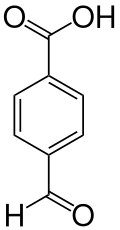
4-Carboxybenzaldehyde
 | |
| Names | |
|---|---|
| Preferred IUPAC name
4-Formylbenzoic acid[1] | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.009.645 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C8H6O3 |
| Molar mass | 150.133 g·mol−1 |
| Appearance | white solid |
| Melting point | 245 °C (473 °F; 518 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
4-Carboxybenzaldehyde (CBA) is an organic compound with the formula OCHC6H4CO2H. It consists of a benzene ring substituted with both an aldehyde and a carboxylic acid, with these functional groups on opposite corners of the ring. This compound is formed in 0.5% yield as a byproduct in the production terephthalic acid from p-xylene. Since approximately 40,000,000 tons of terephthalic acid are produced per year, CBA is a relatively large scale industrial chemical.[2]
Uses
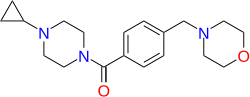
One known use of 4-Carboxybenzaldehyde is in the synthesis of Bavisant (JNJ-31001074) [929622-08-2].[3] Another use is in the synthesis of revefenacin.
See also
- 2-Carboxybenzaldehyde
References
- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. P-65.1.6.3. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ Barnicki, Scott D. (2012). "Synthetic Organic Chemicals". In Kent, James A. (ed.). Handbook of Industrial Chemistry and Biotechnology. Boston, MA: Springer US. pp. 307–389. doi:10.1007/978-1-4614-4259-2_10. ISBN 978-1-4614-4258-5.
- ^ Brett D. Allison, et al. WO2007035425 (to Janssen Pharmaceutica NV).