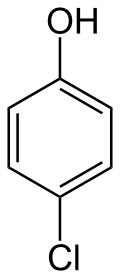
4-Chlorophenol
 | |
| Names | |
|---|---|
| Preferred IUPAC name
4-Chlorophenol | |
| Other names
p-Chlorophenol
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
507004 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.094 |
| EC Number |
|
Gmelin Reference
|
2902 |
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2020 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H5ClO |
| Molar mass | 128.56 g·mol−1 |
| Appearance | White solid |
| Density | 1.2651 g/cm3 at 40 °C[1] |
| Melting point | 43.1 °C (109.6 °F; 316.2 K)[1] |
| Boiling point | 219 °C (426 °F; 492 K)[1] |
Solubility in water
|
27.1 g/L |
| Acidity (pKa) | 9.41[2] |
Magnetic susceptibility (χ)
|
−77.7·10−6 cm3/mol[3] |
Refractive index (nD)
|
1.5579[1] |
| Structure[4] | |
Dipole moment
|
2.11 D |
| Thermochemistry[5] | |
Std enthalpy of
formation (ΔfH⦵298) |
−197.7 kJ·mol−1 (s) −181.3 kJ·mol−1 (l) |
Enthalpy of fusion (ΔfH⦵fus)
|
14.1 kJ·mol−1 |
| Hazards | |
| GHS labelling: | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H290, H301, H302, H312, H314, H332, H411 |
Precautionary statements
|
P234, P260, P264, P270, P271, P273, P280, P301+P310, P301+P312, P301+P330+P331, P302+P352, P303+P361+P353, P304+P312, P304+P340, P305+P351+P338, P310, P312, P321, P322, P330, P363, P390, P391, P404, P405, P501 |
| Flash point | 121 °C (250 °F; 394 K)[6] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
4-Chlorophenol is an organic compound with the formula C6H4ClOH. It is one of three monochlorophenol isomers. It is a colorless or white solid that melts easily and exhibits significant solubility in water. Its pKa is 9.41.
Preparation and reaction
It is prepared by chlorination of phenol, preferably in polar solvents, which tends to yield the 4-chloro derivative. Direct chlorination of molten phenol favors the formation of 2-chlorophenol.[7]
It once was produced on a large scale as a precursor to hydroquinone.[7] It is a classic precursor, upon reaction with phthalic anhydride, to quinizarin.[8] The commercial dye quinizarin is produced by the reaction of phthalic anhydride and 4-chlorophenol followed by hydrolysis of the chloride.[9]
References
- ^ a b c d Haynes, p. 3.116
- ^ Haynes, p. 5.90
- ^ Haynes, p. 3.577
- ^ Haynes, p. 9.61
- ^ Haynes, pp. 5.10, 6.156
- ^ Haynes, p. 16.20
- ^ a b Muller, François; Caillard, Liliane (2011). "Chlorophenols". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a07_001.pub2. ISBN 978-3-527-30673-2.
- ^ Bigelow, L. A.; Reynolds, H. H. (1926). "Quinizarin". Org. Synth. 6: 78. doi:10.15227/orgsyn.006.0078.
- ^ Bien, H.-S.; Stawitz, J.; Wunderlich, K. "Anthraquinone Dyes and Intermediates". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_355. ISBN 978-3-527-30673-2.
Cited sources
- Haynes, William M., ed. (2016). CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. ISBN 9781498754293.