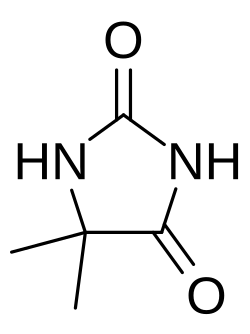
5,5-Dimethylhydantoin
 | |
| Names | |
|---|---|
| IUPAC name
4,4-Dimethyl-2,5-dioxoimidazolidine
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.957 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C5H8N2O2 |
| Molar mass | 128.131 g·mol−1 |
| Boiling point | 178 °C (352 °F; 451 K) |
| Solubility | very soluble in ethanol, ether, acetone, benzene, chloroform soluble in DMSO |
| log P | -0.48 |
| Vapor pressure | 0.0000028 mmHg |
| Acidity (pKa) | 9.19 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
5,5-Dimethylhydantoin (DMH, systematic name 4,4-dimethyl-2,5-dioxoimidazolidine) is a derivative of hydantoin having two methyl groups as substituents on the ring carbon. DMH can be used in silver electroplating baths as an alternative to cyanide.[1] It is a degradation product of 1,3-dibromo-5,5-dimethylhydantoin that can be detected on foods that have not been properly washed.[2] It can be easily produced by a Bucherer–Bergs reaction, warming a mixture of acetone cyanohydrin and ammonium carbonate.[3]
Reaction of DMH with various halogens gives N,N′-dihalo derivatives, such as diido-5,5-dimethylhydantoin,[4][5] bromochloro-5,5-dimethylhydantoin,[6] and dibromo-5,5-dimethylhydantoin, which can be used as reagents to supply of electrophilic halogen atoms or hypohalous acids (or synthetic equivalents of them). Sodium 4,4-dimethyl-2,5-dioxoimidazolidin-1-ide, the sodium salt of its conjugate base, has been commercialized as a stabilizer for active chlorine in the papermaking industry.[7]
See also
- Cyanuric acid, another N-heterocyclic compound used for stabilizing halogens
References
- ^ Liu, Anmin; Ren, Xuefeng; Wang, Chong; Zhang, Jiale; Du, Chunhui; Han, Ruolin; An, Maozhong (2021). "DMH and NA–based cyanide-free silver electroplating bath: A promising alternative to cyanide ones in microelectronics". Ionics. 27: 417–422. doi:10.1007/s11581-020-03541-5.
- ^ Lee, Doo-Sik; Shim, Ji-Won; Shin, Jae-Wook (2025). "Development and validation of an analytical method for the determination of 5,5-dimethylhydantoin in various processed food". Food Science and Biotechnology. 34 (6): 1309–1316. doi:10.1007/s10068-024-01747-1. PMC 11914416. PMID 40110394.
- ^ Wagner, E. C.; Baizer, Manuel (1940). "5,5-Dimethylhydantoin". Organic Syntheses. 20: 42.
- ^ Ricco, Christophe (2013). "1,3-Diiodo-5,5-dimethylhydantoin". Synlett. 24 (16): 2173–2174. doi:10.1055/s-0033-1339477.
- ^ Orazi, Orfeo O.; Corral, Renée A.; Bertorello, Héctor E. (1965). "N-Iodohydantoins. II. Iodinations with 1,3-Diiodo-5,5-dimethylhydantoin". The Journal of Organic Chemistry. 30 (4): 1101–1104. doi:10.1021/jo01015a036.
- ^ Yasukazu Ura, Gozyo Sakata. "Chloroamines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a06_553. ISBN 978-3-527-30673-2.
- ^ "Equinox® Stabilizer: A Highly Effective, Halogen Stabilizer For Wet End Paper Applications".