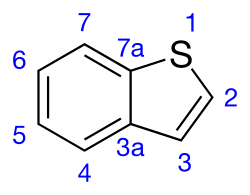
Benzothiophene
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-Benzothiophene | |
| Other names
Benzo[b]thiophene
Thianaphthene Benzothiofuran | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.178 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C8H6S |
| Molar mass | 134.20 g·mol−1 |
| Appearance | White solid |
| Density | 1.15 g/cm3 |
| Melting point | 32 °C (90 °F; 305 K) |
| Boiling point | 221 °C (430 °F; 494 K) |
| Hazards | |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Warning |
Hazard statements
|
H302, H411 |
Precautionary statements
|
P264, P270, P273, P301+P312, P330, P391, P501 |
| Flash point | 110 °C (230 °F; 383 K) |
| Related compounds | |
Related compounds
|
Thiophene, Indene, Benzofuran, Indole |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Benzothiophene is an aromatic organic compound with a molecular formula C8H6S and an odor similar to naphthalene (mothballs). It occurs naturally as a constituent of petroleum-related deposits such as lignite tar. Benzothiophene has no household use. In addition to benzo[b]thiophene, a second isomer is known: benzo[c]thiophene.[3]
Benzothiophene finds use in research as a starting material for the synthesis of larger, usually bioactive structures. It is found within the chemical structures of pharmaceutical drugs such as raloxifene, zileuton, and sertaconazole, and also BTCP. It is also used in the manufacturing of dyes such as thioindigo.
Synthesis
Most syntheses of benzothiophene create substituted benzothiophenes as a precursor to further reactions. An example is the reaction of an alkyne-substituted 2-bromobenzene with either sodium sulfide or potassium sulfide to form benzothiophene with an alkyl substitution at position 2.[4]
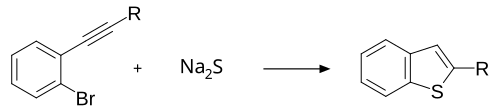
Thiourea can be used as a reagent in place of sodium sulfide or potassium sulfide.[5]
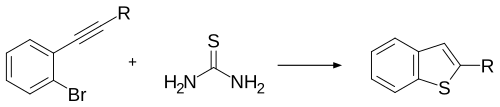
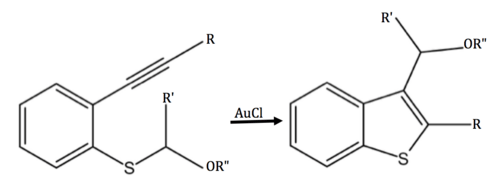
In the presence of a gold catalyst, a more complex 2,3-disubstituted benzothiophene can be synthesised.[6]
References
- ^ Merck Index, 11th Edition, 9232
- ^ "Thianaphthene". www.sigmaaldrich.com. Sigma Aldrich. Retrieved 12 November 2020.
- ^ Cava, Michael P.; Lakshmikantham, M. V. (1975). "Nonclassical Condensed Thiophenes". Accounts of Chemical Research. 8 (4): 139–44. doi:10.1021/ar50088a005.
- ^ Sun, Lei-Lei; Deng, Chen-Liang; Tang, Ri-Yuan; Zhang, Xing-Guo (16 September 2011). "CuI/TMEDA-Catalyzed Annulation of 2-Bromo Alkynylbenzenes with Na2S: Synthesis of Benzo[b]thiophenes". The Journal of Organic Chemistry. 76 (18). American Chemical Society (ACS): 7546–7550. doi:10.1021/jo201081v. ISSN 0022-3263. PMID 21812478.
- ^ Kuhn, Marius; Falk, Florian C.; Paradies, Jan (5 August 2011). "Palladium-Catalyzed C–S Coupling: Access to Thioethers, Benzo[b]thiophenes, and Thieno[3,2-b]thiophenes". Organic Letters. 13 (15). American Chemical Society (ACS): 4100–4103. doi:10.1021/ol2016093. ISSN 1523-7060. PMID 21732682.
- ^ Nakamura, Itaru; Sato, Takuma; Yamamoto, Yoshinori (3 July 2006). "Gold-Catalyzed Intramolecular Carbothiolation of Alkynes: Synthesis of 2,3-Disubstituted Benzothiophenes from (α-Alkoxy Alkyl) (ortho-Alkynyl Phenyl) Sulfides". Angewandte Chemie International Edition. 45 (27). Wiley: 4473–4475. doi:10.1002/anie.200601178. ISSN 1433-7851. PMID 16767784.