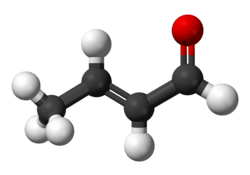
Crotonaldehyde
 | |
 | |
| Names | |
|---|---|
| IUPAC name
(2E)-but-2-enal
| |
| Other names
Crotonaldehyde
Crotonic aldehyde β-Methacrolein β-Methyl acrolein 2-butenal Propylene aldehyde | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.021.846 |
| EC Number |
|
IUPHAR/BPS
|
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII |
|
| UN number | 1143 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties ((E) isomer) | |
Chemical formula
|
C4H6O |
| Molar mass | 70.091 g·mol−1 |
| Appearance | colourless liquid |
| Odor | pungent, suffocating odor |
| Density | 0.846 g/cm3 |
| Melting point | −76.5 °C (−105.7 °F; 196.7 K) |
| Boiling point | 104.0 °C (219.2 °F; 377.1 K) |
Solubility in water
|
18% (20 °C)[2] |
| Solubility | very soluble in ethanol, ethyl ether, acetone soluble in chloroform miscible in benzene |
| Vapor pressure | 19 mmHg (20 °C)[2] |
Refractive index (nD)
|
1.4362 |
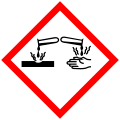
| Hazards | |
| GHS labelling: | |
Pictograms
|
|
Signal word
|
Danger |
Hazard statements
|
H225, H301, H310, H311, H315, H318, H330, H335, H341, H373, H400 |
Precautionary statements
|
P201, P202, P210, P233, P240, P241, P242, P243, P260, P262, P264, P270, P271, P273, P280, P281, P284, P301+P310, P302+P350, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P312, P314, P320, P321, P330, P332+P313, P361, P362, P363, P370+P378, P391, P403+P233, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 
4
3
2 |
| Flash point | 13 °C (55 °F; 286 K) |
Autoignition
temperature |
207 °C (405 °F; 480 K) |
| Explosive limits | 2.1–15.5% |
| Lethal dose or concentration (LD, LC): | |
LC50 (median concentration)
|
600 ppm (rat, 30 min) 1375 ppm (rat, 30 min) 519 ppm (mouse, 2 hr) 1500 ppm (rat, 30 min)[3] |
LCLo (lowest published)
|
400 ppm (rat, 1 hr)[3] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 2 ppm (6 mg/m3)[2] |
REL (Recommended)
|
TWA 2 ppm (6 mg/m3)[2] |
IDLH (Immediate danger)
|
50 ppm[2] |
| Related compounds | |
Related alkenals
|
Acrolein |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Crotonaldehyde is a chemical compound with the formula CH3CH=CHCHO. The compound is usually sold as a mixture of the E- and Z-isomers, which differ with respect to the relative position of the methyl and formyl groups. The E-isomer is more common. This lachrymatory liquid is moderately soluble in water and miscible in organic solvents. As an unsaturated aldehyde, crotonaldehyde is a versatile intermediate in organic synthesis. It occurs in a variety of foodstuffs, e.g. soybean oils.[4]
Production and reactivity
Crotonaldehyde is produced by the aldol condensation of acetaldehyde:
- 2 CH3CHO → CH3CH=CHCHO + H2O
Crotonaldehyde is a multifunctional molecule that exhibits diverse reactivity. It is a prochiral dienophile.[5] It is a Michael acceptor. Addition of methylmagnesium chloride produces 3-penten-2-ol.[6]
Uses
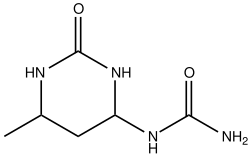
It is a precursor to many fine chemicals. A prominent industrial example is the crossed aldol condensation with diethyl ketone to give trimethylcyclohexenone, this can be easily converted to trimethylhydroquinone, which is a precursor to the vitamin E.[8] Other derivatives include crotonic acid, 3-methoxybutanol and the food preservative Sorbic acid. Condensation with two equivalents of urea gives a pyrimidine derivative that is employed as a controlled-release fertilizer. [4]
Safety
Crotonaldehyde is a potent irritant even at the ppm levels. It is not very toxic, with an LD50 of 174 mg/kg (rats, oral).[4]
See also
References
- ^ Merck Index, 11th Edition, 2599
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0157". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Crotonaldehyde". Immediately Dangerous to Life or Health Concentrations. National Institute for Occupational Safety and Health.
- ^ a b c R. P. Schulz; J. Blumenstein; C. Kohlpaintner (2005). "Crotonaldehyde and Crotonic Acid". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a08_083. ISBN 978-3-527-30673-2.
- ^ Longley Jr., R. I..; Emerson, W. S.; Blardinelli, A. J. (1954). "3,4-Dihydro-2-methoxy-4-methyl-2H-pyran". Org. Synth. 34: 29. doi:10.15227/orgsyn.034.0029.
- ^ Coburn, E. R. (1947). "3-Penten-2-ol". Org. Synth. 27: 65. doi:10.15227/orgsyn.027.0065.
- ^ Dittmar, Heinrich; Drach, Manfred; Vosskamp, Ralf; Trenkel, Martin E.; Gutser, Reinhold; Steffens, Günter (2009). "Fertilizers, 2. Types". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.n10_n01. ISBN 978-3-527-30673-2.
- ^ Müller, Marc-André; Schäfer, Christian; Litta, Gilberto; Klünter, Anna-Maria; Traber, Maret G.; Wyss, Adrian; Ralla, Theo; Eggersdorfer, Manfred; Bonrath, Werner (6 December 2022). "100 Years of Vitamin E: From Discovery to Commercialization" (PDF). European Journal of Organic Chemistry. 2022 (45). doi:10.1002/ejoc.202201190.