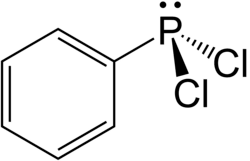
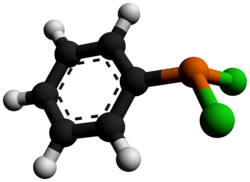
Dichlorophenylphosphine
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Phenylphosphonous dichloride | |
| Other names
Dichlorophenylphosphane
Phenylphosphorus dichloride | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.010.388 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2798 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H5Cl2P |
| Molar mass | 178.98 g·mol−1 |
| Appearance | colorless liquid |
| Odor | acrid, pungent |
| Density | 1.3190 g/mL |
| Melting point | −51 °C (−60 °F; 222 K) |
| Boiling point | 222 °C (432 °F; 495 K) |
Solubility in water
|
insoluble |
| Solubility | miscible in benzene, CS2, chloroform |
Refractive index (nD)
|
1.6030 |
| Hazards | |
| GHS labelling: | |
Pictograms
|
|
Signal word
|
Danger |
Hazard statements
|
H290, H301, H302, H314, H335 |
Precautionary statements
|
P234, P260, P264, P270, P271, P280, P301+P310, P301+P312, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P330, P363, P390, P403+P233, P404, P405, P501 |
| NFPA 704 (fire diamond) | 
3
1
3 W |
| Flash point | 101 °C (214 °F; 374 K) |
Autoignition
temperature |
159 °C (318 °F; 432 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
200 mg/kg (oral, rat) |
| Safety data sheet (SDS) | Fisher MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Dichlorophenylphosphine is an organophosphorus compound with the formula C6H5PCl2. This colourless viscous liquid is commonly used in the synthesis of organophosphines.
Dichlorophenylphosphine is commercially available. It may be prepared by an electrophilic substitution of benzene by phosphorus trichloride, catalyzed by aluminium chloride.[1][2] However, aluminum chloride often induces diarylation; a cleaner catalyst for monoarylation is stannic chloride.[2] The compound is an intermediate for the synthesis of other chemicals for instance dimethylphenylphosphine:
- C6H5PCl2 + 2 CH3MgI → C6H5P(CH3)2 + 2 MgICl
Many tertiary phosphines can be prepared by this route.[3]
In the McCormack reaction dichlorophenylphosphine adds dienes to give the chlorophospholenium ring.[4]
Reductive coupling of the dichlorophosphine gives the cyclophosphine (PhP)5.[5]
References
- ^ B. Buchner; L. B. Lockhart, Jr. (1951). "Phenyldichlorophosphine". Organic Syntheses. 31: 88. doi:10.15227/orgsyn.031.0088.
- ^ a b Engel, Robert; Cohen, Jaime-Lee Iolani (2004). Synthesis of Carbon–Phosphorus Bonds (2 ed.). CRC. §6.2.3. ISBN 0-8493-1617-0.
- ^ P. Loeliger E. Flückiger (1976). "Sulfide Contraction via Alkylative Coupling: 3-Methyl-2,4-heptanedione". Organic Syntheses. 55: 127. doi:10.15227/orgsyn.055.0127.
- ^ W. B. McCormack (1963). "3-Methyl-1-Phenylphospholene oxide". Org. Synth. 43: 73. doi:10.15227/orgsyn.043.0073.
- ^ Marianne Baudler, Klaus Glinka (1993). "Monocyclic and Polycyclic Phosphines". Chem. Rev. 93: 1623–1667. doi:10.1021/cr00020a010.
