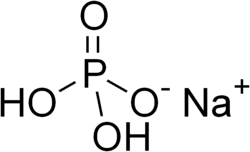
Monosodium phosphate
 | |
 Phosphorus, P Oxygen, O Hydrogen, H Sodium, Na | |
| Names | |
|---|---|
| IUPAC name
Sodium dihydrogen phosphate
| |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.028.591 |
| E number | E339(i) (antioxidants, ...) |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
NaH2PO4 |
| Molar mass | 119.976 g·mol−1 |
| Appearance | White powder or crystals |
| Density | 2.36 g/cm3 (anhydrous) |
Solubility in water
|
59.90 g/(100 mL) (0°C) |
| Hazards | |
| NFPA 704 (fire diamond) | 
0
0
0 |
| Flash point | Non-flammable |
| Related compounds | |
Other cations
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Monosodium phosphate (MSP), also known as monobasic sodium phosphate and sodium dihydrogen phosphate, is an inorganic compound with the chemical formula NaH2PO4. It is a sodium salt of phosphoric acid. It consists of sodium cations (Na+) and dihydrogen phosphate anions (H2PO−4). One of many sodium phosphates, it is a common industrial chemical. The salt exists in an anhydrous form, as well as monohydrate and dihydrate (NaH2PO4·H2O and NaH2PO4·2H2O respectively).[1]
Production and reactions
The salt is obtained by partial neutralization of phosphoric acid.
The pKa of monosodium phosphate is 6.8-7.2 (depending on the physicochemical characteristics during pKa determination).[2]
Heating this salt above 169 °C gives disodium pyrophosphate:
- 2 NaH2PO4 → Na2H2P2O7 + H2O
When heated at 550 °C, anhydrous trisodium trimetaphosphate is formed:[3]
- 3 NaH2PO4 → Na3P3O9 + 3 H2O
Uses
Phosphates are often used in foods and in water treatment. The pH of such formulations is adjusted by mixtures of various sodium phosphates, such as this salt.[1] It is added in animal feed for its nutritional value.[1]
References
- ^ a b c Schrödter, Klaus; Bettermann, Gerhard; Staffel, Thomas; Wahl, Friedrich; Klein, Thomas; Hofmann, Thomas (2008). "Phosphoric Acid and Phosphates". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_465.pub3. ISBN 978-3-527-30673-2.
- ^ Salaun, F.: "Influence of mineral environment on the buffering capacity of casein micelles" Archived 2012-03-13 at the Wayback Machine, "Milchwissenschaft", 62(1):3
- ^ Bell, R. N. (1950). "Sodium Metaphosphates". Inorganic Syntheses. Vol. 3. pp. 103–106. doi:10.1002/9780470132340.ch26.