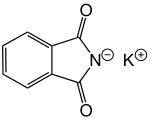
Potassium phthalimide
Potassium phthalimide is a chemical compound of formula C8H4KNO2. It is the potassium salt of phthalimide, and usually presents as fluffy, very pale yellow crystals. It can be prepared by adding a hot solution of phthalimide in ethanol to a solution of potassium hydroxide in ethanol; the desired product precipitates.[1]
 | |
| Identifiers | |
|---|---|
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.012.770 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
C8H4KNO2 |
| Molar mass | 185.221 g/mol |
| Appearance | Light yellow solid |
| Melting point | > 300 °C (572 °F; 573 K) |
Solubility in water |
Soluble in water |
| Hazards | |
| NFPA 704 (fire diamond) | 
1
2
0 |
| Related compounds | |
Related compounds |
Phthalimide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
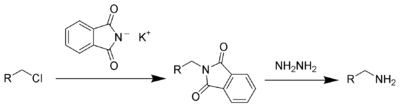
This compound is a commercially available reagent used in the Gabriel synthesis of amines.
References
- P. L. Salzberg and J. V. Supniewski (1941). "β-Bromoethylphthalimide". Organic Syntheses.; Collective Volume, 1, p. 119
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.