Sodium trimetaphosphate
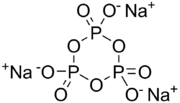
Sodium trimetaphosphate (also STMP), with formula Na3P3O9, is a metaphosphate of sodium. It has the empirical formula NaPO3. It is the sodium salt of trimetaphosphoric acid. It is a colourless solid that finds specialised applications in food and construction industries. STMP is prepared by heating sodium polyphosphate at a few hundred degrees. [2]
 | |
| Names | |
|---|---|
| Other names
Sodium trimetaphosphate | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ECHA InfoCard | 100.029.171 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
Na3P3O9 |
| Molar mass | 305.885 g/mol |
| Appearance | colorless or white crystals |
| Density | 2.49 g/cm3 (anhydrous) 1.786 g/cm3 (hexahydrate) |
| Melting point | 53 °C (127 °F; 326 K) (hexahydrate, decomposes to anyhdrous) |
Solubility in water |
22 g/100 mL |
| Solubility | insoluble in alcohol |
Refractive index (nD) |
1.433 (hexahydrate) |
| Structure | |
Crystal structure |
triclinic (hexahydrate) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Although drawn with a particular resonance structure, the trianion has D3d symmetry.
References
- Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, Florida: CRC Press. pp. 4–86. ISBN 0-8493-0594-2.
- Klaus Schrödter; Gerhard Bettermann; Thomas Staffel; Friedrich Wahl; Thomas Klein; Thomas Hofmann (2008). Phosphoric Acid and Phosphates. Ullmann’s Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_465.pub3.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.