Tris(2-aminoethyl)amine
amine.svg.png) | |
| Names | |
|---|---|
| Preferred IUPAC name
N1,N1-Bis(2-aminoethyl)ethane-1,2-diamine | |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
1739626 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.021.689 |
| EC Number |
|
Gmelin Reference
|
27074 |
| MeSH | Tris(2-aminoethyl)amine |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2922 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H18N4 |
| Molar mass | 146.238 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Ichtyal, ammoniacal |
| Density | 0.976 g/mL (20 °C)[1] |
| Melting point | −16 °C (3 °F; 257 K) |
| Boiling point | 265 °C (509 °F; 538 K) |
Solubility in water
|
Miscible |
| log P | −2.664 |
| Vapor pressure | 3 Pa (at 20 °C) |
Refractive index (nD)
|
1.497[1] |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
−74.3–−72.9 kJ mol−1 |
Std enthalpy of
combustion (ΔcH⦵298) |
−4860.6–−4859.2 kJ mol−1 |
| Hazards | |
| GHS labelling: | |
Pictograms
|
|
Signal word
|
Danger |
Hazard statements
|
H301, H310, H314 |
Precautionary statements
|
P280, P302+P350, P305+P351+P338, P310 |
| Flash point | 113 °C (235 °F; 386 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
|
| Safety data sheet (SDS) | fishersci.com |
| Related compounds | |
Related amines
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Tris(2-aminoethyl)amine is the organic compound with the formula N(CH2CH2NH2)3. This colourless liquid is soluble in water and is highly basic, consisting of a tertiary amine center and three pendant primary amine groups. Tris(2-aminoethyl)amine is commonly abbreviated as tren or TREN. It is used a crosslinking agent in the synthesis of polyimine networks and a tripodal ligand in coordination chemistry.
Supramolecular and polymer derivatives
Tris(2-aminoethyl)amine has been used to prepare molecular capsules and related supramolecular structures.[2][3][4]
Metal complexes
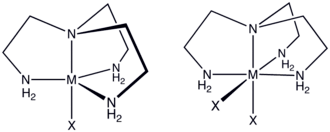
Tren is a C3-symmetric, tetradentate chelating ligand that forms stable complexes with transition metals, especially those in the 2+ and 3+ oxidation states. Tren complexes exist with relatively few isomers, reflecting the constrained connectivity of this tetramine. Thus, only a single achiral stereoisomer exists for [Co(tren)X2]+, where X is halide or pseudohalide.[5] In contrast, for [Co(trien)X2]+ five diastereomers are possible, four of which are chiral. In a few cases, tren serves as a tridentate ligand with one of the primary amine groups non-coordinated. Tren is a common impurity in the more common triethylenetetramine ("trien"). As a trifunctional amine, tren forms a triisocyanate when derivatized with COCl2.
TREN is known to react fast in the presence of (aromatic) aldehydes to form an imine. During this process, water is formed, making it a condensation reaction. Due to this fast and efficient reaction, TREN is commonly used in the preparation of polyimines.[6]
N-methylated derivatives
The permethylated derivative of tren has the formula N(CH2CH2NMe2)3. "Me6tren" forms a variety of complexes but, unlike tren, does not stabilize Co(III). Related amino-triphosphines are also well developed, such as N(CH2CH2PPh2)3 (m.p. 101-102 °C). This species is prepared from the nitrogen mustard N(CH2CH2Cl)3.[7]
N,N,N-trimethyltren, N(CH2CH2NHMe)3 is also available.[8]
Safety considerations
(H2NCH2CH2)3N, like other polyamines, is corrosive.[9] It causes severe skin burns and eye damage, is harmful if inhaled due to the destruction of respiratory tissues, is toxic if swallowed, and can be fatal in contact with skin. Its median lethal dose is 246 mg/kg, oral (rat), and 117 mg/kg, dermal (rabbit). It is also combustible.[10]
References
- ^ a b "Tris(2-aminoethyl)amine". Sigma-Aldrich.
- ^ Denissen, Wim; Rivero, Guadalupe; Nicolaÿ, Renaud; Leibler, Ludwik; Winne, Johan M.; Du Prez, Filip E. (2015). "Vinylogous Urethane Vitrimers". Advanced Functional Materials. 25 (16): 2451–2457. doi:10.1002/adfm.201404553. hdl:11336/2888. S2CID 95154324.
- ^ Akinc, Akin; et al. (2008). "A combinatorial library of lipid-like materials for delivery of RNAi therapeutics". Nature Biotechnology. 26 (5): 561–569. doi:10.1038/nbt1402. PMC 3014085. PMID 18438401.
- ^ Gestwicki, Jason E.; Cairo, Christopher W.; Strong, Laura E.; Oetjen, Karolyn A.; Kiessling, Laura L. (2002). "Influencing Receptor−Ligand Binding Mechanisms with Multivalent Ligand Architecture". Journal of the American Chemical Society. 124 (50): 14922–14933. doi:10.1021/ja027184x. PMID 12475334.
- ^ Donald A. House "Ammonia & N-donor Ligands" in Encyclopedia of Inorganic Chemistry John Wiley & Sons, 2006. doi:10.1002/0470862106.ia009.
- ^ Schoustra, Sybren K.; Dijksman, Joshua A.; Zuilhof, Han; Smulders, Maarten M. J. (2021). "Molecular control over vitrimer-like mechanics – tuneable dynamic motifs based on the Hammett equation in polyimine materials". Chemical Science. 12 (1): 293–302. doi:10.1039/D0SC05458E. PMC 8178953. PMID 34163597.
- ^ R. Morassi, L. Sacconi "Tetradentate Tripod Ligands Containing Nitrogen, Sulfur, Phosphorus, and Arsenic as Donor Atoms" Inorganic Syntheses 1976, vol. 16 p. 174-180. doi:10.1002/9780470132470.ch47
- ^ Schmidt, H.; Lensink, C.; Xi, S. K.; Verkade, J. G. (1989). "New Prophosphatranes: Novel intermediates to five-coordinate phosphatranes". Zeitschrift für Anorganische und Allgemeine Chemie. 578: 75–80. doi:10.1002/zaac.19895780109.
- ^ "The Physical and Theoretical Chemistry Laboratory Oxford University MSDS". Archived from the original on 2011-01-18. Retrieved 2008-04-19.
- ^ "Safety Data Sheet". Sigma-Aldrich. July 1, 2014. Retrieved April 7, 2019.