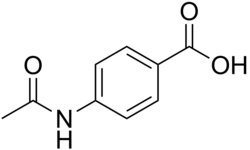
Acedoben
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
4-Acetamidobenzoic acid | |
| Other names
N-Acetyl-PABA; 4-Carboxyacetanilide; p-Acetamidobenzoic acid
p-Acetaminobenzoic acid; PAAB; p-Acetoaminobenzoic acid
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.008.287 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C9H9NO3 |
| Molar mass | 179.175 g·mol−1 |
| Melting point | 259 to 262 °C (498 to 504 °F; 532 to 535 K) (dec.) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Acedoben (4-acetamidobenzoic acid or N-acetyl-PABA) is a chemical compound with the molecular formula of C9H9NO3. It is the acetyl derivative of para-aminobenzoic acid (PABA).
Acedoben, as a salt with dimepranol, is a component of some pharmaceutical preparations such as inosine pranobex.
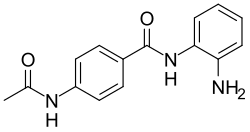
Acedoben has application in the synthesis of a drug that is called Tacedinaline.[3]
See also
- N-Acetylanthranilic acid is an positional isomer of this compound
References
- ^ "4-Acetamidobenzoic acid". Sigma-Aldrich.
- ^ Acedoben, ChemIndustry.com
- ^ Gediya, L. K.; Belosay, A.; Khandelwal, A.; Purushottamachar, P.; Njar, V. C. O. (March 2008). "Improved synthesis of histone deacetylase inhibitors (HDIs) (MS-275 and CI-994) and inhibitory effects of HDIs alone or in combination with RAMBAs or retinoids on growth of human LNCaP prostate cancer cells and tumor xenografts". Bioorganic & Medicinal Chemistry. 16 (6): 3352–3360. doi:10.1016/j.bmc.2007.12.007.