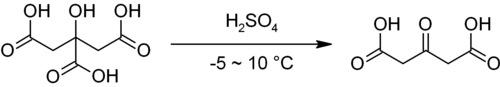Acetonedicarboxylic acid
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
3-Oxopentanedioic acid | |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.007.999 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C5H6O5 |
| Molar mass | 146.09814 g/mol |
| Appearance | colorless or white solid |
| Density | 1.499 g/cm3 |
| Melting point | 122 °C (252 °F; 395 K) (decomposes) |
| Boiling point | 408.4 °C (767.1 °F; 681.5 K) (760mm Hg) |
| Hazards | |
| GHS labelling: | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319, H335 |
Precautionary statements
|
P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 |
| Flash point | 214.9 °C (418.8 °F; 488.0 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
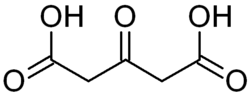
Acetonedicarboxylic acid is the organic compound with the formula O=C(CH2CO2H)2. It is classified as both a dicarboxylic acid and an oxocarboxylic acid.[2]
Preparation
Acetonedicarboxylic acid is prepared commercially by oxidation of citric acid.[2] It can also be prepared by treating citric acid with fuming sulfuric acid.[3]
Reactions and uses
Upon heating it undergoes decarboxylation first to give acetoacetic acid then acetone:[2]
- O=C(CH2CO2H → O=C(CH3)(CO2H) + CO2
- O=C(CH3)(CO2H) → O=C(CH3)2 + CO2
Acetonedicarboxylic acid and its esters such as dimethylacetonedicarboxylate are primarily used as building blocks in the synthesis of heterocycles[4] One example is it use in Robinson's classic synthesis of tropinone. It participates in the Weiss–Cook reaction.[5]
Related compounds
See also
References
- ^ |ALDRICH&N5=SEARCH_CONCAT_PNO|BRAND_KEY&F=SPEC 1,3-Acetonedicarboxylic acid{{|bot=InternetArchiveBot |fix-attempted=yes }} at Sigma-Aldrich (safety data sheet)
- ^ a b c Klingler, Franz Dietrich; Ebertz, Wolfgang (2000). "Oxocarboxylic Acids". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a18_313. ISBN 978-3-527-30385-4.
- ^ Roger Adams; H. M. Chiles; C. F. Rassweiler (1925). "Acetonedicarboxylic Acid". Organic Syntheses. 5: 5. doi:10.15227/orgsyn.005.0005.
- ^ Stanovnik, Branko; Grošelj, Uroš (2010). "Dialkyl Acetone-1,3-Dicarboxylates and their Mono- and bis(Dimethylamino)methylidene Derivatives in the Synthesis of Heterocyclic Systems". Advances in Heterocyclic Chemistry Volume 100. Vol. 100. pp. 145–174. doi:10.1016/S0065-2725(10)10005-1. ISBN 9780123809360.
- ^ Korman, Matthew; Paz, Eric; Franklin, Tylor; Lewandowski, Nicholas R.; Sullivan, Bethany; Imhoff, Andrea M.; Fisher, Luke; Bichler, Katherine A.; Van Ornum, Scott G. (2020). "Process Development of the Weiss–Cook Reaction for the Preparation of cis-1,5-Dimethylbicyclo[3.3.0]octane-3,7-dione in the Undergraduate Organic Laboratory". Journal of Chemical Education. 97 (10): 3835–3838. Bibcode:2020JChEd..97.3835K. doi:10.1021/acs.jchemed.9b00653. S2CID 225248640.