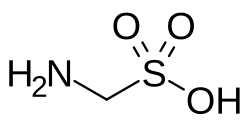
Aminomethylsulfonic acid
 | |
| Names | |
|---|---|
| IUPAC name
Aminomethanesulfonic acid
| |
| Other names
Aminomethylenesulfonic acid
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.034.212 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
CH5NO3S |
| Molar mass | 111.12 g·mol−1 |
| Melting point | 190–194 °C (374–381 °F; 463–467 K)[1] |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Danger |
Hazard statements
|
H314 |
Precautionary statements
|
P260, P264, P280, P301+P330+P331, P302+P361+P354, P304+P340, P305+P354+P338, P316, P321, P363, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Aminomethylsulfonic acid (also called aminomethylenesulfonic acid and aminomethanesulfonic acid) is an organic chemical compound, with amino (-NH2) and sulfonic acid (-SO3H) groups on the carbon atom. Its chemical formula is NH2CH2SO3H, and it has a molar mass of 111.12 g/mol.
Properties
Aminomethanesulfonic acid is a corrosive solid. It could be thought of as the sulfonic acid analogue of glycine, just like aminomethylphosphonic acid is the phosphonic analogue of glycine.[2] Its density is ~1.7 g/cm3.[3]
Synthesis
Aminomethane sulfonic acid can be made by the reaction between hexamethylenetetramine (hexamine) with sulfur dioxide (SO2) in an aqueous environment.[4]
Dangers
Aminomethanesulfonic acid can cause skin and eye burns, it is an dermatotoxin.
References
- ^ "Aminomethanesulfonic acid (CAS RN: 13881-91-9)". CAS Common Chemistry. Chemical Abstracts Service. Retrieved 2026-01-15.
- ^ CID 83791 from PubChem
- ^ CSID:75612, https://www.chemspider.com/Chemical-Structure.75612.html, (accessed 16:39, Jan 15, 2026)
- ^ Khoma, R. E.; et al. (2019). "Synthesis, crystal structure, and spectral characteristics of N-(n-propyl) aminomethanesulfonic acid. Acute toxicity of aminomethanesulfonic acid and its N-alkylated derivatives". Voprosy Khimii i Khimicheskoi Tekhnologii. 6 (6): 255–262. doi:10.32434/0321-4095-2019-127-6-255-262.