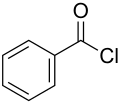
Benzoyl chloride
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Benzoyl chloride | |||
Other names
| |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.002.464 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1736 | ||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
C6H5COCl | ||
| Molar mass | 140.57 g·mol−1 | ||
| Appearance | colorless liquid | ||
| Odor | Benzaldehyde-like, but more pungent | ||
| Density | 1.211 g/mL[1] | ||
| Melting point | −1 °C (30 °F; 272 K)[1] | ||
| Boiling point | 198 °C (388 °F; 471 K)[1] | ||
Solubility in water
|
Reacts. Decomposes to HCl and benzoic acid. | ||
| Vapor pressure | 1 hPa (0.75 mmHg) (32 °C (90 °F; 305 K))[1] | ||
Magnetic susceptibility (χ)
|
−75.8×10−6 cm3/mol | ||
| Viscosity | 0.0012 Pa·s[2] | ||
| Structure | |||
Point group
|
Cs | ||
| Hazards | |||
| GHS labelling:[1] | |||
Pictograms
|
  
| ||
Signal word
|
Danger | ||
Hazard statements
|
H227, H302+H312, H314, H317, H331, H402 | ||
Precautionary statements
|
P210, P261, P264, P270, P271, P272, P273, P280, P301+P312+P330, P301+P330+P331, P303+P361+P353, P304+P340+P310, P305+P351+P338+P310, P333+P313, P363, P370+P378, P403+P233, P403+P235, P405, P501 | ||
| NFPA 704 (fire diamond) | 
3
2
2 W | ||
| Flash point | 72 °C (162 °F; 345 K)[1] | ||
Autoignition
temperature |
600 °C (1,112 °F; 873 K)[1] | ||
| Explosive limits | 2.5% to 27%(V)[1] | ||
Threshold limit value (TLV)
|
0.5 ppm[2] (C) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
|||
LC50 (median concentration)
|
3.03 mg/L (Inhalation, rat, 4h)[2] | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Benzoyl chloride, also known as benzenecarbonyl chloride, is an organochlorine compound with the formula C6H5COCl. It is a colourless, fuming liquid with an irritating odour, and consists of a phenyl ring (C6H5) with an acyl chloride (−C(=O)Cl) substituent. It is mainly useful for the production of organic peroxides but is also used in other areas such as in the preparation of dyes, perfumes, pharmaceuticals, and resins.[3]
Preparation
Benzoyl chloride is produced from benzotrichloride using either water or benzoic acid:[3]
- C6H5CCl3 + H2O → C6H5COCl + 2 HCl
- C6H5CCl3 + C6H5CO2H → 2 C6H5COCl + HCl
As with other acyl chlorides, it can be generated from the parent acid and standard chlorinating agents such as phosphorus pentachloride, thionyl chloride, and oxalyl chloride. It was first prepared by treatment of benzaldehyde with chlorine.[4]
An early method for production of benzoyl chloride involved chlorination of benzyl alcohol.[5]
Reactions
It reacts with water to produce hydrochloric acid and benzoic acid:
- C6H5COCl + H2O → C6H5COOH + HCl
Benzoyl chloride is a typical acyl chloride. It reacts with alcohols to give the corresponding esters. Similarly, it reacts with amines to give the amide.[6][7]
It undergoes the Friedel-Crafts acylation with aromatic compounds to give the corresponding benzophenones and related derivatives.[8] With carbanions, it serves again as a source of the benzoyl cation synthon, C6H5CO+.[9]
Benzoyl peroxide, a common reagent in polymer chemistry, is produced industrially by treating benzoyl chloride with hydrogen peroxide and sodium hydroxide:[10]
- 2 C6H5COCl + H2O2 + 2 NaOH → (C6H5CO)2O2 + 2 NaCl + 2 H2O
Modified benzoyl chlorides
Many substituted derivatives of benzoyl chloride are known. 4-Nitrobenzoyl chloride is a precursor to the anesthetic procaine.[3] One example is 4-chlorobenzoyl chloride, a precursor to 4-chloro-4'-hydroxybenzophenone, which is used to make high performance polymers.[11]
Safety
Benzoyl chloride is toxic and a serious skin irritant.[3]
See also
References
- ^ a b c d e f g h Sigma-Aldrich Co., Benzoyl chloride. Retrieved on 3 January 2026.
- ^ a b c d e f "Benzoyl chloride SDS". thermofisher.com. ThermoFisher. 21 December 2025. p. 5. Retrieved 3 January 2026.
- ^ a b c d Maki, Takao; Takeda, Kazuo (2000). "Benzoic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_555. ISBN 978-3-527-30673-2.
- ^ Wöhler, Friedrich; von Liebig, Justus (1832). "Untersuchungen über das Radikal der Benzoesäure". Annalen der Pharmacie (in German). 3 (3): 262–266. doi:10.1002/jlac.18320030302. hdl:2027/hvd.hxdg3f.
- ^ US patent 1851832, Wilmot, H.B. & Garnett, P.J., "Manufacture and production of benzoyl chloride", published 1932-03-29, issued 1932-03-29
- ^ Marvel, C. S.; Lazier, W. A. (1929). "Benzoyl Piperidine". Organic Syntheses. 9: 16. doi:10.15227/orgsyn.009.0016; Collected Volumes, vol. 1, 1941, p. 99.
- ^ Saha, P.; Ali, M. A.; Punniyamurthy, T. (2011). "Ligand-free Copper(ii) Oxide Nanoparticles Catalyzed Synthesis Of Substituted Benzoxazoles". Organic Syntheses. 88: 398–405. doi:10.15227/orgsyn.088.0398.
- ^ Minnis, W. (1932). "Phenyl Thienyl Ketone". Organic Syntheses. 12: 62. doi:10.15227/orgsyn.012.0062; Collected Volumes, vol. 2, 1943.
- ^ Fujita, M.; Hiyama, T. (1990). "Phenyl Thienyl Ketone". Organic Syntheses. 69: 44. doi:10.15227/orgsyn.069.0044; Collected Volumes, vol. 8, 1993.
- ^ El-Samragy, Yehia (2004). "Chemical and Technical Assessment". Benzoyl Peroxide (PDF). 61st JECFA (Technical report). Joint FAO/WHO Expert Committee on Food Additives. p. 1. Retrieved 31 October 2013.
Benzoyl peroxide is synthesized commercially by the reaction of benzoyl chloride, sodium hydroxide, and hydrogen peroxide. Traces of benzoic acid remain after usual purification procedures.
- ^ Parker, David; Bussink, Jan; Van De Grampel, Hendrik T.; Wheatley, Gary W.; Dorf, Ernst-Ulrich; Ostlinning, Edgar; Reinking, Klaus; Schubert, Frank; Jünger, Oliver; Wagener, Reinhard (2012). "Polymers, High-Temperature". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_449.pub4. ISBN 978-3-527-30673-2.