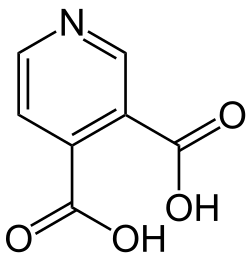
Cinchomeronic acid
 | |
| Names | |
|---|---|
| Preferred IUPAC name
pyridine-3,4-dicarboxylic acid | |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
137242 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.006 |
| EC Number |
|
Gmelin Reference
|
487254 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C7H5NO4 |
| Molar mass | 167.120 g·mol−1 |
| Appearance | pale yellow crystalline |
| Melting point | 256 °C (493 °F; 529 K) |
Solubility in water
|
sparingly soluble |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319, H335 |
Precautionary statements
|
P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Cinchomeronic acid is an organic compound with the molecular formula C7H5NO4.[1] The acid was initially isolated by Hugo Weidel in 1868.[2] The compound is one of six isomers of pyridinedicarboxylic acid that are dicarboxylic derivatives of pyridine.[3]
Synthesis
Cinchomeronic acid is obtained as a degradation product of various Cinchona alkaloids. For example, it is obtained from quinine, cinchonidine, cinchonine, and apoquinine using nitric acid or by oxidation of β-collidine, lepidine, or isoquinoline.[4][5]
Cinchomeric acid can also be prepared by oxidation of isoquinoline with potassium permanganate.[6]
Physical properties
The acid forms white to pale yellow crystalline compound derived from Cinchona alkaloids. It demonstrates low solubility in water, but is more soluble in organic solvents.
It exhibits dimorphism. A conventional orthorhombic structure (called "Form I") and a newly found primitive monoclinic structure ("Form II", space group P21/c) exist.[7]
Uses
It is utilized as a precursor in the synthesis of pharmaceutical compounds and as a ligand in coordination chemistry.
References
- ^ a b PubChem. "Cinchomeronic acid". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-01-25.
- ^ Die wissenschaftliche Welt von gestern: die Preisträger des Ignaz L. Lieben-Preises 1865 - 1937 und des Richard Lieben-Preises 1912 - 1928 ; ein Kapitel österreichischer Wissenschaftsgeschichte in Kurzbiografien. Wien Köln Weimar: Böhlau. 2004. p. 59. ISBN 978-3-205-77303-0. Retrieved 24 January 2026.
- ^ Bernthsen, August (1889). A Text-book of Organic Chemistry. D. Van Nostrand Company. p. 488. Retrieved 24 January 2026.
- ^ Wolffenstein, Richard (1922). Die Pflanzenalkaloide (3rd ed.). Berlin, Heidelberg: Springer Berlin Heidelberg. p. 66. ISBN 978-3-642-92449-1. Retrieved 24 January 2026.
{{cite book}}: ISBN / Date incompatibility (help) - ^ Journal of the Chemical Society. Chemical Society (Great Britain). 1879. p. 947. Retrieved 24 January 2026.
- ^ Breitmaier, Eberhard; Jung, Günther (2014). Organische Chemie, 7. vollst. Überarb. u. erw. Auflage 2012: Grundlagen,Verbindungsklassen, Reaktionen, Konzepte, Molekülstruktur, Naturstoffe, Syntheseplanung, Nachhaltigkeit (7., vollständig überarbeitete und erweiterte Auflage ed.). Stuttgart: Thieme. p. 706. ISBN 978-3-13-159987-2.
- ^ Evans, Ivana Radosavljevic; Howard, Judith A. K.; Evans, John S. O.; Postlethwaite, Stella R.; Johnson, Mark R. (8 September 2008). "Polymorphism and hydrogen bonding in cinchomeronic acid: a variable temperature experimental and computational study". CrystEngComm. 10 (10): 1404–1409. Bibcode:2008CEG....10.1404E. doi:10.1039/B807015F. ISSN 1466-8033. Retrieved 24 January 2026.