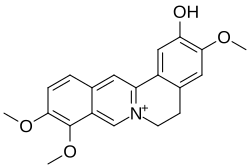
Columbamine
 | |
| Names | |
|---|---|
| IUPAC name
3,9,10-Trimethoxy-5,6-dihydroisoquinolino[2,1-b]isoquinolin-7-ium-2-ol
| |
| Other names
Dehydroisocorypalmine
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C20H20NO4+ |
| Molar mass | 338.382 g·mol−1 |
| Melting point | 280–282 °C (536–540 °F; 553–555 K) (chloride salt)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Columbamine is an isoquinoline alkaloid made up of four rings, two of which contain nitrogen. It is related to berberine but without its methylenedioxy group. Columbamine is also called dehydroisocorypalmine.[2] It has usually been isolated and characterised as its chloride salt.[1]
Occurrence
Columbamine has been found in several Berberis species, Coptis chinensis[3] and Jateorhiza palmata.[1]
Biosynthesis
The biosynthesis of columbamine starts with tyrosine and proceeds via (S)-reticuline in a pathway leading to benzylisoquinoline alkaloids.[4][5] The final step is catalysed by the enzyme tetrahydroberberine oxidase, which oxidises (S)-isocorypalmine:[1][6]
References
- ^ a b c d Mascavage, Linda M.; Jasmin, Serge; Sonnet, Philip E.; Wilson, Michael; Dalton, David R. (2010). "Alkaloids". Ullmann's Encyclopedia of Industrial Chemistry. p. 54. doi:10.1002/14356007.a01_353.pub2. ISBN 978-3-527-30385-4.
- ^ PubChem. "Columbamine". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-04-29.
- ^ Wu-Chang Chuang; Dun-Shyurng Young; Lilian Kao Liu; Shuenn-Jyi Sheu (1996). "Liquid chromatographic-electrospray mass spectrometric analysis of Coptidis Rhizoma". Journal of Chromatography A. 755: 19–26. doi:10.1016/S0021-9673(96)00591-2.
- ^ Tian, Ya; Kong, Lingzhe; Li, Qi; Wang, Yifan; Wang, Yongmiao; An, Zhoujie; Ma, Yuwei; Tian, Lixia; Duan, Baozhong; Sun, Wei; Gao, Ranran; Chen, Shilin; Xu, Zhichao (2024). "Structural diversity, evolutionary origin, and metabolic engineering of plant specialized benzylisoquinoline alkaloids". Natural Product Reports. 41 (11): 1787–1810. doi:10.1039/d4np00029c. PMID 39360417.
- ^ Begley, Tadhg P. (2009). Encyclopedia of Chemical Biology. Vol. 10. Wiley. pp. 1569–1570. doi:10.1002/cbic.200900262. ISBN 978-0-471-75477-0.
- ^ Enzyme 1.3.3.8 at KEGG Pathway Database.
-Isocorypalmine.svg.png)