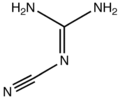
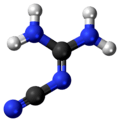
2-Cyanoguanidine
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
N′′-Cyanoguanidine | |||
Other names
| |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
| ||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.006.649 | ||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
C2H4N4 | ||
| Molar mass | 84.082 g·mol−1 | ||
| Appearance | White crystals | ||
| Density | 1.4 g/cm3 | ||
| Melting point | 209.5 °C (409.1 °F; 482.6 K) | ||
| Boiling point | 252 °C (486 °F; 525 K) | ||
Solubility in water
|
41.3 g/L[1] | ||
| Solubility in acetone | soluble | ||
| Solubility in ethanol | soluble | ||
| log P | −1.5 (est)[2] | ||
Henry's law
constant (kH) |
2.25×10−10 (atm·m3)/mol | ||
| Acidity (pKa) | 14.8[1] | ||
| Basicity (pKb) | 14.4[1] | ||
Magnetic susceptibility (χ)
|
−44.55×10−6 cm3/mol | ||
| Hazards[4] | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
>30000 mg/kg (oral, rat)[3] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
2-Cyanoguanidine is a nitrile derived from guanidine. It is a dimer of cyanamide, from which it can be prepared.
Production and use
2-Cyanoguanidine is produced by treating cyanamide with base. It is produced in soil by decomposition of cyanamide. A variety of useful compounds are produced from 2-cyanoguanidine, guanidines and melamine. For example, acetoguanamine and benzoguanamine are prepared by condensation of cyanoguanidine with the nitrile:[5][6]
- (H2N)2C=NCN + PhCN → 1,3-(H2N)-5-Ph(C3N3)
Cyanoguanidine is also used as a nitrification inhibitor in stabilized nitrogen fertilizers.[1] It is used in the adhesive industry as a curing agent for epoxy resins.[1] Formerly, it was used as a fuel in some explosives, and may be used as a stabilizer for nitrocellulose.[7]
It was patented for use in the synthesis of methylphenobarbital.[8]
Chemistry
The structure has been confirmed by X-ray crystallography.[9] The molecule is planar with two primary amine groups attached to an imino nitrile. According to Raman spectroscopy measurements, solutions also contain a tautomer, (H2N)HN=CN(H)CN.[10]
2-cyanoguanidine is isomeric to ammonium dicyanamide, from which it may be prepared by heating in a closed vessel.[1][11] The reaction proceeds via a zwitterionic intermediate, analogous to the Wöhler synthesis of urea from ammonium cyanate.[11]
References
- ^ a b c d e f Güthner, Thomas; Mertschenk, Bernd. "Cyanamides". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a08_139.pub2. ISBN 978-3-527-30673-2.
- ^ International Chemical Safety Card 0650
- ^ Sakai, akira (30 January 2004). SIDS Initial Assessment Report, Cyanoguanidine (PDF). SIAM 17. Arona, Italy: UNEP Publications. Archived from the original (PDF) on 2017-05-17. Retrieved 24 October 2025.
- ^ Sigma-Aldrich Co., Dicyandiamide.
- ^ Deim, H.; Matthias, G.; Wagner, R. A. "Amino Resins". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a02_115.pub2. ISBN 978-3-527-30673-2.
- ^ Simons, J. K.; Saxton, M. R. (1953). "Benzoguanamine". Org. Synth. 33: 13. doi:10.15227/orgsyn.033.0013; Coll. Vol., vol. 4, 1963, p. 78.
- ^ Fedoroff, Basil T.; Sheffield, Oliver E. (1 January 1966). Encyclopedia of Explosives and Related Items: Volume 3 - Chlorides through Detonating Relays (PDF). Dover, NJ: Picatinny Arsenal. p. C-587. AD0653029. Retrieved 25 October 2025.
- ^ Ludwig Taub and Walter Kropp, U.S. patent 2,061,114 (1936 to Winthrop Chemical Company Inc.).
- ^ Hirshfeld, F. L.; Hope, H. (1980). "An X-ray determination of the charge deformation density in 2-cyanoguanidine". Acta Crystallographica Section B Structural Crystallography and Crystal Chemistry. 36 (2): 406–415. Bibcode:1980AcCrB..36..406H. doi:10.1107/S0567740880003366.
- ^ Alı́a, J.M.; Edwards, H.G.M.; Garcı́a Navarro, F.J. (2001). "The imino–amino tautomeric equilibrium in cyanoguanidine dissolved in several aprotic solvents; an FT-Raman spectroscopic study". Journal of Molecular Structure. 597 (1–3): 49–55. Bibcode:2001JMoSt.597...49A. doi:10.1016/S0022-2860(01)00579-8.
- ^ a b Lotsch, Bettina V.; Senker, Jürgen; Schnick, Wolfgang (1 February 2004). "Characterization of the Thermally Induced Topochemical Solid-State Transformation of NH4[N(CN)2] into NCN=C(NH2)2 by Means of X-ray and Neutron Diffraction as Well as Raman and Solid-State NMR Spectroscopy" (PDF). Inorganic Chemistry. 43 (3): 895–904. doi:10.1021/ic034984f. Archived from the original (PDF) on 11 August 2017.