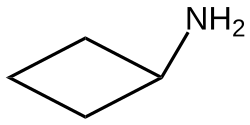
Cyclobutylamine
 | |
| Names | |
|---|---|
| Other names
aminocyclobutane
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.017.942 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C4H9N |
| Molar mass | 71.123 g·mol−1 |
| Appearance | colorless liquid |
| Boiling point | 80.5–81.5 °C (176.9–178.7 °F; 353.6–354.6 K) |
Refractive index (nD)
|
1.4356 |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H225, H314 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P260, P264, P280, P301+P330+P331, P302+P361+P354, P303+P361+P353, P304+P340, P305+P354+P338, P316, P321, P363, P370+P378, P403+P235, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Cyclobutylamine is an organic compound with the formula C4H7NH2. It is a colorless, readily distillable liquid. It can be prepared from cyclobutanecarboxylic acid via the amide or by Curtius rearrangement.[1] Cyclobutylamine is a member of the aminocycloalkanes, which also includes cyclopropylamine, cyclopentylamine, and cyclohexylamine.
Oxidation with potassium permanganate gives cyclobutanone.[2]
Using "strain-relief reaction", substituted bicyclobutanes can be converted to a variety of N-substituted cyclobutylamines.[3]
References
- ^ Newton W. Werner, Joseph Casanova, Jr. (1967). "Cyclobutylamine". Organic Syntheses. 47: 28. doi:10.15227/orgsyn.047.0028.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Lee, Donald G.; Ribagorda, María; Adrio, Javier (2007). "Potassium Permanganate". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/9780470842898.rp244.pub2. ISBN 978-0-471-93623-7.
- ^ Gianatassio, Ryan; Lopchuk, Justin M.; Wang, Jie; Pan, Chung-Mao; Malins, Lara R.; Prieto, Liher; Brandt, Thomas A.; Collins, Michael R.; Gallego, Gary M.; Sach, Neal W.; Spangler, Jillian E.; Zhu, Huichin; Zhu, Jinjiang; Baran, Phil S. (2016). "Strain-release amination". Science. 351 (6270): 241–246. Bibcode:2016Sci...351..241G. doi:10.1126/science.aad6252. PMC 4730898. PMID 26816372.
Wikimedia Commons has media related to Cyclobutylamine.