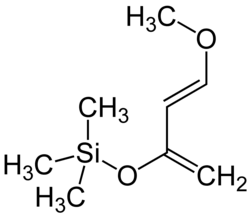
Danishefsky's diene
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
{[(3E)-4-Methoxybuta-1,3-dien-2-yl]oxy}trimethylsilane | |
| Other names
Kitahara diene
trans-1-Methoxy-3-trimethylsilyloxy-buta-1,3-diene (E)-1-Methoxy-3-trimethylsilyloxy-buta-1,3-diene | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.157.252 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C8H16O2Si |
| Molar mass | 172.299 g·mol−1 |
| Density | 0.89 g cm−3 (20 °C)[2] |
| Boiling point | 68 to 69 °C (154 to 156 °F; 341 to 342 K) at 0.0189 kPa[2] |
| Hazards | |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Warning |
Hazard statements
|
H226, H315, H319 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P264, P280, P302+P352, P303+P361+P353, P305+P351+P338, P321, P332+P313, P337+P313, P362, P370+P378, P403+P235, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Danishefsky's diene (Kitahara diene) is an organosilicon compound and a diene with the formal name trans-1-methoxy-3-trimethylsilyloxy-buta-1,3-diene named after Samuel J. Danishefsky.[3][4] Because the diene is very electron-rich it is a very reactive reagent in Diels-Alder reactions. This diene reacts rapidly with electrophilic alkenes, such as maleic anhydride. The methoxy group promotes highly regioselective additions. The diene is known to react with amines,[5] aldehydes, alkenes and alkynes.[4] Reactions with imines [6] and nitro-olefins [7] have been reported.
It was first synthesized by the reaction of trimethylsilyl chloride with 4-methoxy-3-buten-2-one and zinc chloride:[8]
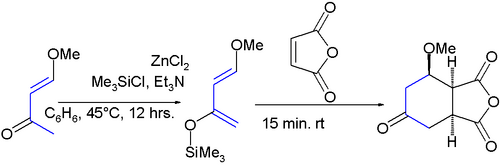
The diene has two features of interest: the substituents promote regiospecific addition to unsymmetrical dienophiles and the resulting adduct is amenable to further functional group manipulations after the addition reaction. High regioselectivity is obtained with unsymmetrical alkenes with a preference for a 1,2-relation of the ether group with the electron-deficient alkene-carbon. All this is exemplified in this aza Diels-Alder reaction:[9][10]
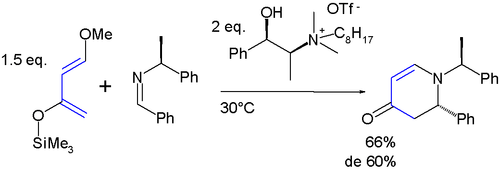
In the cycloaddition product, the silyl ether is a synthon for a carbonyl group through the enol. The methoxy group is susceptible to an elimination reaction enabling the formation of a new alkene group.
Applications in asymmetric synthesis have been reported.[11][12][13][14][15][16][17][18][19][20][21][22] Derivatives have been reported.[23]
References
- ^ Sigma-Aldrich product page.
- ^ a b Sicherheitsdatenblatt Merck.
- ^ Danishefsky, Samuel J.; Kitahara, T. (1974). "Useful diene for the Diels-Alder reaction". J. Am. Chem. Soc. 96: 7807–7808. doi:10.1021/ja00832a031.
- ^ a b Strategic Applications of Named Reactions in Organic Synthesis, Laszlo Kurti,Barbara Czako, Elsevier 2005
- ^ Yuan, Yu (2002). "Acid-Free Aza Diels−Alder Reaction of Danishefsky's Diene with Imines". Organic Letters. 4: 3309–3311. doi:10.1021/ol0265822.
- ^ Loncaric, Catherine (2003). "Alkaline salt-catalyzed aza Diels–Alder reactions of Danishefsky's diene with imines in water under neutral conditions". Chemical Communications (5): 574–575. doi:10.1039/B300880K.
- ^ Node, Manabu (1996). "Exo selective Diels?Alder reaction of nitroolefins with Danishefsky's diene". Chemical Communications (22): 2559. doi:10.1039/CC9960002559.
- ^ Preparation and Diels-Alder Reaction of a Highly Nucleophilic Diene. Org. Synth., Coll. Vol. 7, p.312 (1990); Vol. 61, p.147 (1983). Link
- ^ Asymmetric aza-Diels-Alder reaction of Danishefsky's diene with imines in a chiral reaction medium Pegot B, Nguyen Van Buu O, Gori D, Vo-Thanh G Beilstein Journal of Organic Chemistry, 2006 Link Archived 2006-10-11 at the Wayback Machine
- ^ This is an asymmetric reaction with a chiral ionic liquid as chiral solvent. The reported chemical yield is 66% with 60% diastereomeric excess
- ^ Simonsen, KB; Svenstrup, N; Roberson, M; Jorgensen, KA (2000). "Development of an unusually highly enantioselective hetero-Diels-Alder reaction of benzaldehyde with activated dienes catalyzed by hypercoordinating chiral aluminum complexes". Chemistry. 6: 123–8. PMID 10747395.
- ^ Yao, S; Saaby, S; Hazell, RG; Jorgensen, KA (2000). "Catalytic enantioselective aza-Diels-alder reactions of imines--an approach to optically active nonproteinogenic alpha-amino acids". Chemistry. 6: 2435–48. PMID 10939745.
- ^ Han, G; LaPorte, MG; Folmer, JJ; Werner, KM; Weinreb, SM (October 2000). "Total syntheses of the Securinega alkaloids (+)-14,15-dihydronorsecurinine, (-)-norsecurinine, and phyllanthine". J. Org. Chem. 65: 6293–306. PMID 11052071.
- ^ Janey, JM; Iwama, T; Kozmin, SA; Rawal, VH (2000). "Racemic and asymmetric Diels-Alder reactions of 1-(2-oxazolidinon-3-yl)-3-siloxy-1,3-butadienes". J Org Chem. 65: 9059–68. PMID 11149852.
- ^ Bernardelli, Patrick (2001). "Total Asymmetric Synthesis of the Putative Structure of the Cytotoxic Diterpenoid (−)-Sclerophytin A and of the Authentic Natural Sclerophytins A and B". Journal of the American Chemical Society. 123: 9021–9032. doi:10.1021/ja011285y.
- ^ Wang, Jing (2001). "A Straightforward Stereoselective Synthesis of d - and l -5-Hydroxy-4-hydroxymethyl-2-cyclohexenylguanine". The Journal of Organic Chemistry. 66: 8478–8482. doi:10.1021/jo015924z.
- ^ Yamashita, Yasuhiro (2003). "Chiral Hetero Diels−Alder Products by Enantioselective and Diastereoselective Zirconium Catalysis. Scope, Limitation, Mechanism, and Application to the Concise Synthesis of (+)-Prelactone C and (+)-9-Deoxygoniopypyrone". Journal of the American Chemical Society. 125: 3793–3798. doi:10.1021/ja028186k.
- ^ Du, Haifeng (2003). "Enantioselective Catalysis of Hetero Diels−Alder Reaction and Diethylzinc Addition Using a Single Catalyst". Organic Letters. 5: 1091–1093. doi:10.1021/ol034143c.
- ^ Ghosh, Arun K. (1996). "Asymmetric hetero Diels-Alder reactions of Danishefsky's diene and glyoxylate esters catalyzed by chiral bisoxazoline derived catalysts". Tetrahedron: Asymmetry. 7: 2165–2168. doi:10.1016/0957-4166(96)00261-3.
- ^ Zhao, Bei (2013). "Asymmetric Hetero-Diels–Alder Reaction of Danishefsky's Dienes with α-Carbonyl Esters Catalyzed by an Indium(III)–PyBox Complex". Organic Letters. 15: 2914–2917. doi:10.1021/ol400841s.
- ^ Wang, Bin (2002). "A Highly Enantioselective Hetero-Diels−Alder Reaction of Aldehydes with Danishefsky's Diene Catalyzed by Chiral Titanium(IV) 5,5',6,6',7,7',8,8'-Octahydro-1,1'-bi-2-naphthol Complexes". The Journal of Organic Chemistry. 67: 2175–2182. doi:10.1021/jo016240u.
- ^ Zheng, Jianfeng (2014). "Asymmetric Hetero-Diels-Alder Reaction of Danishefsky's Diene with α-Ketoesters and Isatins Catalyzed by a Chiral N , N′ -Dioxide/Magnesium(II) Complex". Chemistry - A European Journal. 20: 14493–14498. doi:10.1002/chem.201404144.
- ^ Amii, Hideki (2001). "Difluorinated Danishefsky's Diene: A Versatile C 4 Building Block for the Fluorinated Six-Membered Rings". Organic Letters. 3: 3103–3105. doi:10.1021/ol0163631.