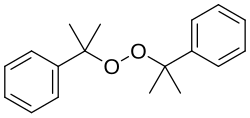
Dicumyl peroxide
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,1′-[peroxydi(propane-2,2-diyl)]dibenzene | |
| Other names
DCUP
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.164 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 3110 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C18H22O2 |
| Molar mass | 270.372 g·mol−1 |
| Appearance | colorless solid |
| Density | 1.062 g/cm3 |
| Melting point | 39 °C (102 °F; 312 K) |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H242, H315, H319, H360, H411 |
Precautionary statements
|
P203, P210, P234, P240, P264, P264+P265, P273, P280, P302+P352, P305+P351+P338, P318, P321, P332+P317, P337+P317, P362+P364, P370+P378, P391, P403, P405, P410, P411, P420, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Dicumyl peroxide is an organic compound with the formula (C6H5CMe2O)2 (Me = CH3). Classified as a dialkyl peroxide, it is produced on a large scale industrially for use in polymer chemistry. It serves as an initiator and crosslinking agent in the production of low density polyethylene.[2]
Production
It is synthesized as a by-product in the autoxidation of cumene, which mainly affords cumene hydroperoxide. Alternatively, it can be produced by the addition of hydrogen peroxide to α-methylstyrene.
Of the approximately 60,000 ton/y production of dialkyl peroxides, dicumyl peroxide is dominant.[3]
Properties
Dicumyl peroxide is relatively stable compound owing to the steric protection provided by the several substituents adjacent to the peroxide group. Upon heating, it breaks down by homolysis of the relatively weak O-O bond.
References
- ^ "Dicumyl peroxide". pubchem.ncbi.nlm.nih.gov.
- ^ Stevens, Malcolm P. (1993). "Polymer Additives: III. Surface Property and Processing Modifiers". Journal of Chemical Education. 70 (9): 713. Bibcode:1993JChEd..70..713S. doi:10.1021/ed070p713.
- ^ Klenk, Herbert; Götz, Peter H.; Siegmeier, Rainer; Mayr, Wilfried. "Peroxy Compounds, Organic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_199.pub2. ISBN 978-3-527-30673-2.