Dihydrobiopterin
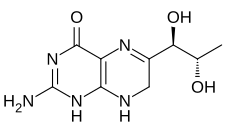 L-erythro-7,8-dihydrobiopterin
| |
| Names | |
|---|---|
| IUPAC name
2-Amino-6-(1,2-dihydroxypropyl)-7,8-dihydro-1H-pteridin-4-one
| |
| Other names
7,8-Dihydrobiopterin
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C9H13N5O3 |
| Molar mass | 239.235 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Dihydrobiopterin (BH2) is a pteridine compound produced in the synthesis of L-DOPA, dopamine, serotonin, norepinephrine and epinephrine. It is restored to the required cofactor tetrahydrobiopterin by dihydrobiopterin reductase.
Biochemistry
Dihydrobiopterin is an intermediate in the pathway to the cofactor, tetrahydrobiopterin, which is used by three aromatic amino acid hydroxylase enzymes which are important in the metabolism of the amino acid phenylalanine and to make the neurotransmitters serotonin, melatonin, dopamine, noradrenaline, and adrenaline.[1]
In higher organisms, tetrahydrobiopterin is derived from guanosine triphosphate by the action of a sequence of three enzymes GTP cyclohydrolase, 6-pyruvoyltetrahydropterin synthase and sepiapterin reductase.[2] The final enzyme performs a stepwise reduction of L-sepiapterin
In some bacteria, for example Chlorobium tepidum, the reduction of L-sepiapterin leads to the L-threo isomer of dihydrobiopterin.[3]
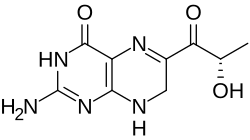
Quinonoid isomer
When tetrahydrobiopterin is used in oxidation reactions as an enzyme cofactor, it can be converted into an alternative isomer of the L-erythro-7,8-dihydrobiopterin form of dihydrobiopterin that is involved in the biosynthesis. This compound, called (6R)-L-erythro-6,7-dihydrobiopterin, is unstable and may spontaneously revert to the more stable isomer. Nevertheless, the enzyme 6,7-dihydropteridine reductase acts directly on it to allow its recycling to tetrahydrobiopterin:[4][5]
See also
- Pteridine
- Dihydropteridine reductase deficiency
References
- ^ Kappock, T. Joseph; Caradonna, John P. (1996). "Pterin-Dependent Amino Acid Hydroxylases". Chemical Reviews. 96 (7): 2659–2756. doi:10.1021/CR9402034. PMID 11848840.
- ^ Werner, Ernst R.; Blau, Nenad; Thöny, Beat (2011). "Tetrahydrobiopterin: Biochemistry and pathophysiology". Biochemical Journal. 438 (3): 397–414. doi:10.1042/BJ20110293. PMID 21867484.
- ^ Cho, Seung-Hyun; Na, Jong-Uk; Youn, Hwan; Hwang, Cheol-Sang; Lee, Chang-Hun; Kang, Sa-Ouk (1999). "Sepiapterin reductase producing l-threo-dihydrobiopterin from Chlorobium tepidum". Biochemical Journal. 340 (2): 497–503. doi:10.1042/bj3400497.
- ^ Lind, Kirsten E. (1972). "Dihydropteridine Reductase". European Journal of Biochemistry. 25 (3): 560–562. doi:10.1111/j.1432-1033.1972.tb01728.x.
- ^ Kono, Haruka; Hara, Satoshi; Furuta, Tadaomi; Ichinose, Hiroshi (2023). "Binding profile of quinonoid -dihydrobiopterin to quinonoid -dihydropteridine reductase examined by in silico and in vitro analyses". The Journal of Biochemistry. 174 (5): 441–450. doi:10.1093/jb/mvad062. PMID 37540845.
-Tetrahydrobiopterin_structure.svg.png)
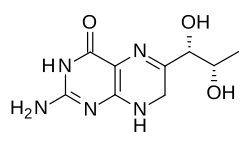
-L-erythro-6%252C7-dihydrobiopterin.svg.png)