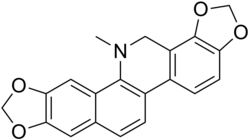
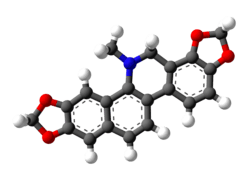
Dihydrosanguinarine
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
13-Methyl-13,14-dihydro-2H,10H-[1,3]benzodioxolo[5,6-c][1,3]dioxolo[4,5-i]phenanthridine | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C20H15NO4 |
| Molar mass | 333.343 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Dihydrosanguinarine is an alkaloid found in the herbs Corydalis adunca and Lamprocapnos spectabilis.[1] It gets its name from the Sanguinaria canadensis plant in the poppy family Papaveraceae in which it is also found.[2]
Biosynthesis
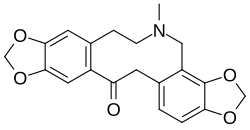
Dihydrosanguinarine is a benzylisoquinoline alkaloid produced in plants from the amino acid tyrosine. A sequence of enzyme-catalysed reactions gives (S)-norcoclaurine which goes through three further transformations to yield (S)-3'-hydroxy-N-methylcoclaurine. (S)-reticuline is made when the enzyme 3'-hydroxy-N-methyl-(S)-coclaurine 4'-O-methyltransferase adds a methyl group. Reticuline is a branch point in the biosynthesis: the (R) enantiomer is used to eventually produce morphine, for example in the opium poppy Papaver somniferum while other pathways use the initially-formed (S) isomer leading to sanguinarine via dihydrosanguinarine.[3]
The immediate precursor to dihydrosanguinarine is protopine, which is oxidised by protopine 6-monooxygenase to 6-hydroxyprotopine, a compound which spontaneously forms the final ring system.[4]
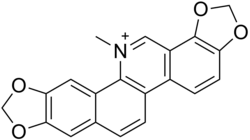
The last step in the sequence to sanguinarine is catalysed by the enzyme dihydrobenzophenanthridine oxidase:[5][2]
See also
- Dihydrosanguinarine 10-monooxygenase
- Epidemic dropsy
References
- ^ Israilov, I. A.; Melikov, F. M.; Murav'Eva, D. A. (1984). "Alkaloids of Dicentra". Chemistry of Natural Compounds. 20: 74–76. doi:10.1007/BF00574796.
- ^ a b Ignatov, Atanas; Clark, W.Gregg; Cline, Steven D.; Psenak, Mikulas; Krueger, Robert J.; Coscia, Carmine J. (1996). "Elicitation of dihydrobenzophenanthridine oxidase in Sanguinaria canadensis cell cultures". Phytochemistry. 43 (6): 1141–1144. doi:10.1016/S0031-9422(96)00540-7. PMID 8987906.
- ^ Tian, Ya; Kong, Lingzhe; Li, Qi; Wang, Yifan; Wang, Yongmiao; An, Zhoujie; Ma, Yuwei; Tian, Lixia; Duan, Baozhong; Sun, Wei; Gao, Ranran; Chen, Shilin; Xu, Zhichao (2024). "Structural diversity, evolutionary origin, and metabolic engineering of plant specialized benzylisoquinoline alkaloids". Natural Product Reports. 41 (11): 1787–1810. doi:10.1039/d4np00029c. PMID 39360417.
- ^ Tanahashi, Takao; Zenk, Meinhart H. (1990). "Elicitor induction and characterization of microsomal protopine-6-hydroxylase, the central enzyme in benzophenanthridine alkaloid biosynthesis". Phytochemistry. 29 (4): 1113–1122. doi:10.1016/0031-9422(90)85414-B.
- ^ Rho, Denis; Chauret, Nathalie; Laberge, Nathalie; Archambault, Jean (1992). "Growth characteristics of Sanguinaria canadensis L. Cell suspensions and immobilized cultures for production of benzophenanthridine alkaloids". Applied Microbiology and Biotechnology. 36 (5): 611–617. doi:10.1007/BF00183237. PMID 1368065.