Diisopropylmercury
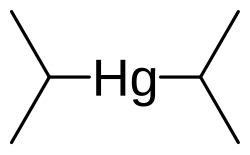 | |
| Names | |
|---|---|
| Preferred IUPAC name
Di(propan-2-yl)mercury | |
| Other names
Bis(1-methylethyl)mercury
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H14Hg |
| Molar mass | 286.770 g·mol−1 |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LDLo (lowest published)
|
7800 μg/kg (intraperitoneal, rat) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Diisopropylmercury is an organomercury chemical compound with formula C6H14Hg.[1] It may be prepared through a reaction between 2-bromopropane and mercury(II) chloride, catalyzed by magnesium. Decomposition of diisopropylmercury by addition of ozone yields a mixture of mercury(II) chloride, mercury(I) chloride, and mercury(II) oxide.[2]
References
- ^ "Bis(1-methylethyl)mercury". PubChem. National Center for Biotechnology Information. Retrieved January 6, 2026.
- ^ Bailey, Philip S., ed. (June 1, 1972). "The Ozonolysis of Organomercurials". Ozone Reactions with Organic Compounds. Advances in Chemistry. Vol. 112. WASHINGTON, D. C.: AMERICAN CHEMICAL SOCIETY. pp. 78–100. doi:10.1021/ba-1972-0112.ch007. ISBN 978-0-8412-0152-1.