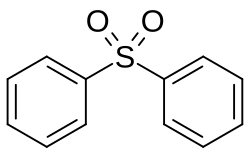
Diphenyl sulfone
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,1′-Sulfonyldibenzene | |
| Other names
Diphenyl sulphone
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.004.413 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C12H10O2S |
| Molar mass | 218.27 g·mol−1 |
| Melting point | 123 °C (253 °F; 396 K) |
| Boiling point | 379 °C (714 °F; 652 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Diphenyl sulfone is an organosulfur compound with the formula (C6H5)2S2. It is a white solid that is soluble in organic solvents. It is a simple example of a molecule with a sulfone functional group.
Uses
It is used as a high temperature solvent. Such high temperature solvents are useful for processing highly rigid polymers, such as PEEK, which only dissolve in very hot solvents.[1]
Preparation and reactions
Diphenyl sulfone is produced by the sulfonation of benzene with sulfuric acid and oleum. For typical processes, benzenesulfonic acid is an intermediate.[2] It is also produced from benzenesulfonyl chloride and benzene.[3]
Oxidation of diphenylsulfide with 30% hydrogen peroxide efficiently gives the sulfone in the presence of a tungstate catalyst.[4]
- (C6H5)2S + 2 H2O2 → (C6H5)2SO2 + 2 H2O
Diphenyl sulfone reacts with butyllithium to give the dilithio derivative:
- (C6H5)2SO2 + 2 C4H9Li → (C6H4Li)2SO2 + 2 C4H10
The dilithio derivative can be quenched with a variety of electrophiles[5]
References
- ^ Parker, David; Bussink, Jan; Van De Grampel, Hendrik T.; Wheatley, Gary W.; Dorf, Ernst-Ulrich; Ostlinning, Edgar; Reinking, Klaus; Schubert, Frank; Jünger, Oliver; Wagener, Reinhard (2012). "Polymers, High-Temperature". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a21_449.pub4. ISBN 978-3-527-30673-2.
- ^ Otto Lindner, Lars Rodefeld "Benzenesulfonic Acids and Their Derivatives" in Ullmann's Encyclopedia of Industrial Chemistry 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a03_507
- ^ Roger Adams, C. S. Marvel, H. T. Clarke, G. S. Babcock, and T. F. Murray (1921). "Benzenesulfonyl chloride". Organic Syntheses. 1: 21
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 1, p. 84. - ^ Sato, Kazuhiko; Hyodo, Mamoru; Aoki, Masao; Zheng, Xiao-Qi; Noyori, Ryoji (2001). "Oxidation of sulfides to sulfoxides and sulfones with 30% hydrogen peroxide under organic solvent- and halogen-free conditions". Tetrahedron. 57 (13): 2469–2476. doi:10.1016/s0040-4020(01)00068-0.
- ^ Chopra, Anu; Dorton, Donna C.; Ogle, Craig A. (1997). "Directed Ortho Dilithiation of Diaryl Compounds: Facile Synthesis of Tricyclic Compounds Based on Heterocycles Fused to Two Benzene Rings". Main Group Metal Chemistry. 20 (12): 783–786. doi:10.1515/mgmc.1997.20.12.783.