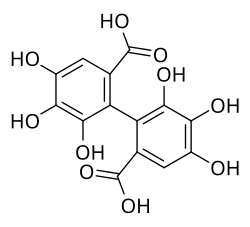
Hexahydroxydiphenic acid
Chemical structure of hexahydroxydiphenic acid
Names
Preferred IUPAC name
4,4′,5,5′,6,6′-Hexahydroxy[1,1′-biphenyl]-2,2′-dicarboxylic acid
Other names
HHDP
Identifiers
CAS Number
3D model (JSmol)
ChemSpider
InChI=1S/C14H10O10/c15-5-1-3(13(21)22)7(11(19)9(5)17)8-4(14(23)24)2-6(16)10(18)12(8)20/h1-2,15-20H,(H,21,22)(H,23,24)
N Key: MFTSECOLKFLUSD-UHFFFAOYSA-N
N InChI=1/C14H10O10/c15-5-1-3(13(21)22)7(11(19)9(5)17)8-4(14(23)24)2-6(16)10(18)12(8)20/h1-2,15-20H,(H,21,22)(H,23,24)
Key: MFTSECOLKFLUSD-UHFFFAOYAZ
O=C(O)c2cc(O)c(O)c(O)c2c1c(O)c(O)c(O)cc1C(=O)O
Properties
Chemical formula
C 14 H 10 O 10
Molar mass
−1
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Hexahydroxydiphenic acid is an organic compound with the formula [(HO)3 C6 HCO2 H]2 . It is the oxidatively coupled derivative of gallic acid [ 2]
Occurrence
Ellagic acid .Luteic acid and ellagic acid are the mono- and dilactone of hexahydroxydiphenic acid, respectively. Hexahydroxydiphenic acid is a component of some ellagitannins,[ 3]
See also
References
^ "MetaCyc hexahydroxydiphenic acid" . biocyc.org .^ Haslam, E.; Cai, Y. (1994). "Plant polyphenols (vegetable tannins): Gallic acid metabolism". Natural Product Reports . 11 (1): 41– 66. doi:10.1039/NP9941100041 . PMID 15206456 . ^ Feldman, Ken S.; Iyer, Malliga R.; Liu, Yanze (2003). "Ellagitannin Chemistry. Studies on the Stability and Reactivity of 2,4-HHDP-Containing Glucopyranose Systems". The Journal of Organic Chemistry . 68 (19): 7433– 7438. doi:10.1021/jo034495x . PMID 12968897 .
Types of ellagitannins
Moieties
Diphenolic acid : Hexahydroxydiphenic acid (HHDP)
Triphenolic acids : Nonahydroxytriphenic acid
Sanguisorbic acid
Valoneic acid
Tetraphenolic acids : Isoterchebulic acid
Terchebulinic acid Lactones
Ellagic acid Gallagic acid
Gallagic acid dilactone
Luteic acid
Sanguisorbic acid dilactone
Valoneic acid dilactone Monomers
Acetonyl geraniin
Alnusiin
Bicornin
Carlesiin
Casuarictin
Emblicanin A and B
Euscaphinin
Galloyl pedunculagin
Grandinin
Helioscopinin B
Jolkinin
Lagerstannin A, B and C
Macranganin
Myrobalanitannin
Nupharin A, B, C, D, E and F
Pedunculagin
Punicalagin
Punigluconin
Phyllanemblinin A, B, C, D, E and F
Punicalin
Roburin E
Rugosin E
Sanguiin H-5
Stenophyllanin A, B and C
Strictinin
Tellimagrandin I and II
Teracatain
Terchebulin
Terflavin A and B
Tergallic acid
Tergallic acid dilactone C-glycosidic ellagitannins
Castalagin (Vescalagin)
Castalin
Casuariin
Casuarinin
Punicacortein A, B, C and D Dehydroellagitannins (molecules with
Alnicortin
Euphorbin A
Euphorscopin
Excoecarianin
Geraniin
Granatin A and B
Helioscopinin A
Mallotusinic acid
Supinanin
Terchebin Transformed ellagitannins
molecules with chebulic acid
Chebulagic acid
Chebulinic acid molecules with Elaeocarpusinic acid
Elaeocarpusin
Helioscopin B
Mallojaponin (1-O-Galloyl-2,4-elaeocarpusinoyl-3,6-(R)-valoneayl-beta-D-glucose)
Oligomers
Dimers
Agrimoniin
Cornusiin E (dimer of tellimagrandin II)
Lambertianin A and B
Nobotanin B
Roburin A, B, C and D
Sanguiin H-6 Trimers
Lambertianin C
Raspberry ellagitannin Tetramers
Lambertianin D
Nobotanin S Pentamer
Melastoflorin A Other
Flavono-ellagitannins (hybrid type)
Urolithins (metabolites)
Pomegranate ellagitannins Category