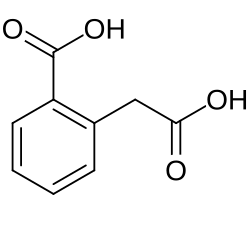
Homophthalic acid
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-(Carboxymethyl)benzoic acid | |
| Other names
α-Carboxy-o-toluic acid
Carboxyphenyl acetic acid | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.740 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C9H8O4 |
| Molar mass | 180.159 g·mol−1 |
| Appearance | Off-white to light yellow or pale green |
| Melting point | 181 °C (358 °F; 454 K)[2] |
| Hazards | |
| GHS labelling: | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319, H335 |
Precautionary statements
|
P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Homophthalic acid is a dicarboxylic acid with the formula C6H4(CO2H)CH2CO2H. It is a colorless solid. The compounds can be prepared by the Willgerodt reaction from 2-acetylbenzoic acid.[3]
One of the uses is in the preparation of the NSAID tesicam.
Further reading
- Use of homophthalic anhydride: Manoni, Francesco; Connon, Stephen J. (2014). "Catalytic Asymmetric Tamura Cycloadditions". Angewandte Chemie International Edition. 53 (10): 2628–2632. doi:10.1002/anie.201309297. PMID 24573916.
References
- ^ "Compound Record".
{{cite web}}: CS1 maint: deprecated archival service (link) - ^ "Homophthalic Acid". www.chemspider.com.
- ^ Schwenk, Erwin; Papa, Domenick (1946). "Preparation of Aryl Aliphatic Acids by the Modified Willgerodt Reaction". The Journal of Organic Chemistry. 11 (6): 798–802. doi:10.1021/jo01176a023. PMID 20282506.