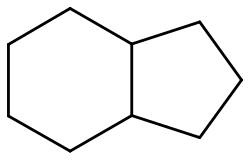
Hydrindane
 | |
| Names | |
|---|---|
| IUPAC name
2,3,3a,4,5,6,7,7a-octahydro-1H-indene
| |
| Other names
octahydroindene, hexahydroindane, bicyclo[4.3.0]nonane
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
2321743 |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.007.104 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C9H16 |
| Molar mass | 124.227 g·mol−1 |
| Density | 0.90732 g/cm3 |
| Melting point | −53 °C (−63 °F; 220 K) |
| Boiling point | 165.5–167.5 °C (329.9–333.5 °F; 438.6–440.6 K) |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
 
|
Signal word
|
Warning |
Hazard statements
|
H226, H302, H413 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P264, P270, P273, P280, P301+P317, P303+P361+P353, P330, P370+P378, P403+P235, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Hydrindane is an organic compound with the formula C9H16. A bicyclic molecule, it is the hydrogenated derivative of the more common hydrocarbons indane and indene. Isomers of hydrindane include the compound with cis-fused rings and the chiral trans-fused derivative.[1]
Occurrence
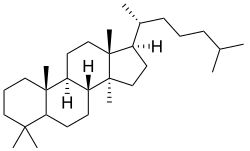
Hydrindane is a component of diesel fuels produced by hydrocracking.[2] Hydrindane is a subunit of many steroids.[3]
References
- ^ Norman L. Allinger, James L. Coke (1960), "The Relative Stabilities of cis and trans Isomers. VII. The Hydrindanes 1,2", Journal of the American Chemical Society, vol. 82, no. 10, pp. 2553–2556, Bibcode:1960JAChS..82.2553A, doi:10.1021/ja01495a039
- ^ McVicker, G. (2002). "Selective Ring Opening of Naphthenic Molecules". Journal of Catalysis. 210 (1): 137–148. Bibcode:2002JCat..210..137M. doi:10.1006/jcat.2002.3685.
- ^ Hans Beyer, Wolfgang Walter (1978), Lehrbuch der organischen Chemie (18. ed.), Stuttgart: S. Hirzel Verlag, p. 350, ISBN 3-7776-0342-2