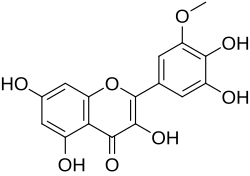
Laricitrin
Chemical structure of laricitrin
Names
IUPAC name
3,3′,4′,5,7-Pentahydroxy-5′-methoxyflavone
Systematic IUPAC name
2-(3,4-Dihydroxy-5-methoxyphenyl)-3,5,7-trihydroxy-4H -1-benzopyran-4-one
Other names
3'-O -Methylmyricetin
Identifiers
CAS Number
3D model (JSmol)
ChEBI
ChemSpider
KEGG
UNII
InChI=1S/C16H12O8/c1-23-11-3-6(2-9(19)13(11)20)16-15(22)14(21)12-8(18)4-7(17)5-10(12)24-16/h2-5,17-20,22H,1H3
N Key: CFYMYCCYMJIYAB-UHFFFAOYSA-N
N InChI=1/C16H12O8/c1-23-11-3-6(2-9(19)13(11)20)16-15(22)14(21)12-8(18)4-7(17)5-10(12)24-16/h2-5,17-20,22H,1H3
Key: CFYMYCCYMJIYAB-UHFFFAOYAK
O=C1c3c(O/C(=C1/O)c2cc(O)c(O)c(OC)c2)cc(O)cc3O
Properties
Chemical formula
C 16 H 12 O 8
Molar mass
−1
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Laricitrin is an O -methylated flavonol, a type of flavonoid. It is found in red grape (absent in white grape)[ 1] Vaccinium uliginosum (bog bilberries).[ 2] [ 3]
Laricitrin is formed from myricetin by the action of the enzyme myricetin O-methyltransferase.[ 4] syringetin .
Glycosides
Laricitrin 3-O-galactoside, found in grape[ 1]
Laricitrin 3-glucoside found in Larix sibirica [ 5]
Laricitrin 3,5’-di-O-β-glucopyranoside, found in Medicago littoralis [ 6]
References
^ a b Mattivi, F; Guzzon, R; Vrhovsek, U; Stefanini, M; Velasco, R (October 2006). "Metabolite profiling of grape: Flavonols and anthocyanins". J. Agric. Food Chem . 54 (20): 7692– 702. Bibcode:2006JAFC...54.7692M . doi:10.1021/jf061538c . PMID 17002441 . ^ Anja; Jaakola, Laura; Riihinen, Kaisu R.; Kainulainen, Pirjo S. (2010). "Anthocyanin and Flavonol Variation in Bog Bilberries (Vaccinium uliginosumL.) in Finland". Journal of Agricultural and Food Chemistry . 58 (1): 427– 433. Bibcode:2010JAFC...58..427L . doi:10.1021/jf903033m . PMID 20000402 . ^ Castillo-Munoz, Noelia; Gomez-Alonso, Sergio; Garcia-Romero, Esteban; Hermosin-Gutierrez, Isidro (2007). "Flavonol profiles of Vitis vinifera red grapes and their single-cultivar wines" Journal of Agricultural and Food Chemistry . 55 (3): 992– 1002. Bibcode:2007JAFC...55..992C . doi:10.1021/jf062800k . PMID 17263504 . ^ Syringetin biosynthesis pathway on metacyc.org ^ Tyukavkina, N. A.; Medvedeva, S. A.; Ivanova, S. Z. (1974). "New flavonol glycosides from the needles of Larix sibirica". Chemistry of Natural Compounds . 10 (2): 170– 172. Bibcode:1974CNatC..10..170T . doi:10.1007/BF00563605 . ^ Flavonoids isolated from Medicago littoralis Rhode (Fabaceae): their ecological and chemosystematic significance Archived 2011-07-22 at the Wayback Machine
Flavonols and their conjugates
Backbone
Flavonols
Aglycones Conjugates
Glycosides of herbacetin Glycosides of kaempferol
Afzelin (Kaempferol 3-rhamnoside)
Astragalin (kaempferol 3-O-glucoside)Kaempferitrin (kaempferol 3,7-dirhamnoside)
Juglanin (Kaempferol 3-O-arabinoside)
Kaempferol 3-alpha-L-arabinopyranoside
Kaempferol 3-alpha-D-arabinopyranoside
Kaempferol 7-alpha-L-arabinoside
Kaempferol 7-O-glucoside
Kaempferol 3-lathyroside
Kaempferol 4'-rhamnoside
Kaempferol 5-rhamnoside
Kaempferol 7-rhamnoside
Kaempferol 7-O-alpha-L-rhamnofuranoside
Kaempferol 3-xyloside
Kaempferol 7-xyloside
Robinin (kaempferol-3-O-robinoside-7-O-rhamnoside)Kaempferol 3-O-rutinoside
Sophoraflavonoloside (Kaempferol 3-O-sophoroside)
Trifolin (Kaempferol 3-O-beta-D-galactoside) Glycosides of myricetin
Betmidin (Myricetin 3-O-arabinoside)
Myricetin 3-O-rutinoside
Myricetin-3-O-neohesperidoside
Myricitrin (Myricetin 3-O-rhamnoside) Conjugates of quercetin
Sulfates Glycosides
Avicularin (quercetin-3-O-α-L-arabinofuranoside)
CTN-986
Guaijaverin (quercetin 3-O-arabinoside )
Heliosin (quercetin 3-digalactoside)
Hyperoside (quercetin 3-O-galactoside)
Isoquercetin (quercetin 3-O-glucoside)Miquelianin (quercetin 3-O-glucuronide)
Quercetin 3,4'-diglucoside
Quercetin-3-sophorodide
Quercitrin (quercetin 3-O-rhamnoside)Rutin (quercetin rutinoside)Reinutrin (quercetin-3-D-xyloside)
Spiraeoside (quercetin 4'-O-glucoside)
Taxillusin (galloylated 3-O-glucoside of quercetin°
O -Methylated flavonols
Aglycones
5-O-methylmyricetin
Annulatin
Ayanin
Axillarin
Azaleatin
Brickellin
Centaureidin
Chrysosplenetin
Combretol
Ermanin
Eupatolitin
Eupalitin
Europetin
Isorhamnetin Jaceidin
Kaempferide Kumatakenin
Laricitrin Natsudaidain
Ombuin
Pachypodol
Patuletin
Retusin
Mearnsetin
Rhamnazin
Rhamnetin Santin
Spinacetin
Syringetin Tamarixetin Glycosides
of isorhamnetin
Narcissin (Isorhamnetin 3-O-rutinoside)
Isorhamnetin 3-O-glucoside
Tamarixetin 7-rutinoside other
Azalein (Azaleatin 3-O-α-L-rhamnoside)
Centaurein (Centaureidin 7-O-glucoside)
Eupalin (Eupalitin 3-0-rhamnoside)
Eupatolin (Eupatolitin 3-O-rhamnoside)
Jacein (Jaceidin 7-O-glucoside)
Patulitrin (Patuletin 7-O-glucoside
Xanthorhamnin (Rhamnetin glycoside)
Derivative flavonols
Aglycones
Noricaritin
Dihydronoricaritin Glycosides
Amurensin
Icariin
Phelloside
Dihydrophelloside
Rutin S
Pyranoflavonols
Furanoflavonols
Semisynthetic
Category