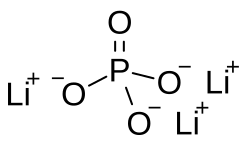
Lithium phosphate
 | |
| Names | |
|---|---|
| IUPAC name
Lithium phosphate
| |
| Other names
Trilithium phosphate
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.030.736 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Li3PO4 |
| Molar mass | 115.794 g/mol |
| Appearance | White powder |
| Density | 2.46 g/cm3[1] |
| Melting point | 1,205 °C (2,201 °F; 1,478 K)[1] |
Solubility in water
|
0.027 g/100 mL (25 °C)[1] |
| Structure[2] | |
Crystal structure
|
Orthorhombic |
Space group
|
Pmn21 |
Lattice constant
|
a = 6.115, b = 5.239, c = 4.855
|
Coordination geometry
|
Tetrahederal (at all atoms) |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
-2095.8 kJ/mol[1] |
| Hazards | |
| GHS labelling: | |
Pictograms
|

|
Signal word
|
Warning |
| NFPA 704 (fire diamond) | 
2
0
0 |
| Related compounds | |
Other cations
|
Trisodium phosphate Tripotassium phosphate Caesium phosphate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Lithium phosphate is a phosphate salt of lithium, with the molecular formula Li3PO4.[3] It is a white solid slightly soluble in water.
It is primarily used in the production of lithium iron phosphate (LiFePO4) for making lithium-ion batteries.[3]
Synthesis and structure
Trilithium phosphate can be produced by the neutralization of lithium carbonate using phosphoric acid:[1]
- 3 Li2CO3 + 2 H3PO4 → 2 Li3PO4 + 3 H2O + 3 CO2
The low-temperature polymorph (β-Li3PO4) crystallizes as an orthorhombic crystal with the space group Pmn21. If heated to above 500 °C, it converts to another polymorph, γ-Li3PO4.[4]
Hazards
Health
Large doses of lithium phosphate may cause dizziness, and sometimes, kidney damage. According to some reports, dehydration, weight loss, and thyroid disturbances can occur due to high exposure of lithium ions. Nervous system defects, including slurred speech, blurred vision, sensory loss, and convulsions may also occur.[3]
See also
- Lithium iron phosphate
- Lithium-ion battery
References
- ^ a b c d e Haynes, W. M., ed. (2016). CRC Handbook of Chemistry and Physics (96th ed.). Boca Raton, Florida: CRC Press/Taylor & Francis. ISBN 978-1482260960.
- ^ Charles Keffer; Alan D. Mighell; Floyd Mauer; Howard E. Swanson; Stanley Block (1967). "Crystal structure of twinned low-temperature lithium phosphate". Inorganic Chemistry. 6 (1): 119–125. doi:10.1021/ic50047a027.
- ^ a b c "Lithium Phosphate". Sigma Aldrich. Retrieved 16 May 2025.
- ^ L. Popović; B. Manoun; D. de Waal; M. K. Nieuwoudt; J. D. Comins (2002). "Raman spectroscopic study of phase transitions in Li3PO4". Journal of Raman Spectroscopy. 34 (1): 77–83. doi:10.1002/jrs.954.