Magnesium silicide
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Magnesium silicide | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.041.125 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 2624 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Mg2Si |
| Molar mass | 76.695 g·mol−1 |
| Appearance | Gray cubic crystals[1] |
| Density | 1.99 g cm−3[1] |
| Melting point | 1,102 °C (2,016 °F; 1,375 K)[1] |
Solubility in water
|
reacts[1] |
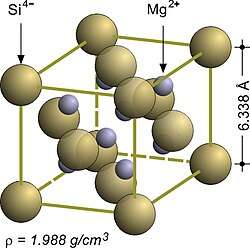
| Structure[2] | |
Crystal structure
|
Antifluorite (cubic), cF12 |
Space group
|
Fm3m, #225 |
Lattice constant
|
a = 0.6351 nm
|
Formula units (Z)
|
4 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
reacts with water to produce pyrophoric silane |
| GHS labelling: | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H261 |
Precautionary statements
|
P231+P232, P280, P370+P378, P402+P404, P501 |
| Related compounds | |
Other cations
|
Calcium silicide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Magnesium silicide, Mg2Si, is an inorganic compound of magnesium and silicon. As-grown Mg2Si usually forms black crystals; they are semiconductors with n-type conductivity and have potential applications in thermoelectric generators.[3]
Crystal structure
Mg2Si crystallizes in the antifluorite structure. In the face-centered cubic lattice, Si centers occupy the corners and face-centered positions of the unit cell, and Mg centers occupy eight tetrahedral sites in the interior of the unit cell. The coordination numbers of Si and Mg are eight and four, respectively.[2]
Synthesis
It can be produced by heating silicon dioxide, SiO2, found in sand, with excess magnesium. The process first forms elemental silicon and magnesium oxide:
- 2 Mg + SiO2 → 2 MgO + Si
If an excess of Mg is present, Mg2Si is formed from the reaction of the remaining magnesium with the elemental silicon:
- 2 Mg + Si → Mg2Si
- If there is an excess of SiO2, then elemental silicon remains.
These reactions proceed exothermically,[4] even explosively.[5]
Reactions
Magnesium silicide can be viewed as consisting of Si4− ions. As such, it is reactive toward acids. Thus, when magnesium silicide is treated with hydrochloric acid, silane (SiH4) and magnesium chloride are produced:
- Mg2Si + 4 HCl → SiH4 + 2 MgCl2
Sulfuric acid can be used as well. These protonolysis reactions are typical of a group 2 (alkaline earth metal) and group 1 (alkali metal) silicides. The early development of silicon hydrides relied on this reaction.[5]
Uses
Magnesium silicide is used to create aluminium alloys of the 6000 series, containing up to approximately 1.5% Mg2Si. An alloy of this group can be age-hardened to form Guinier-Preston zones and a very fine precipitate, resulting in increased alloy strength.[6]
Magnesium silicide is a narrow-gap semiconductor. Its as-grown crystal exhibits n-type conductivity, but it can be changed to p-type by doping with Ag, Ga, Sn and possibly Li (at high doping levels). The major potential electronic application of Mg2Si is in thermoelectric generators.[3][7]
References
- ^ a b c d Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, Florida: CRC Press. p. 4.74. ISBN 1-4398-5511-0.
- ^ a b Noda Y., Kon H., Furukawa Y., Otsuka N., Nishida I.A., Masumoto K. (1992). "Preparation and Thermoelectric Properties of Mg2Si1−xGex (x=0.0~0.4) Solid Solution Semiconductors". Mater. Trans., JIM. 33 (9): 845–850. doi:10.2320/matertrans1989.33.845.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Hirayama, Naomi (2019). "Substitutional and interstitial impurity p-type doping of thermoelectric Mg2Si: a theoretical study". Sci. Technol. Adv. Mater. 20 (1): 160–172. Bibcode:2019STAdM..20..160H. doi:10.1080/14686996.2019.1580537. PMC 6419642. PMID 30891103.

- ^ Ehrlich, P. (1963). "17". In Brauer, Georg (ed.). Handbook of Preparative Inorganic Chemistry (PDF). Vol. 1. Translated by Riley; Reed F. (2nd ed.). New York: Academic Press, Inc. p. 921. ISBN 978-0121266011. Retrieved 26 September 2024.
{{cite book}}: ISBN / Date incompatibility (help) - ^ a b Stock, Alfred; Somieski, Carl (1916). "Siliciumwasserstoffe. I. Die aus Magnesiumsilicid und Säuren entstehenden Siliciumwasserstoffe". Berichte der Deutschen Chemischen Gesellschaft. 49: 111–157. doi:10.1002/cber.19160490114.
- ^ "Properties and Selection: Non-ferrous Alloys and Special Purpose Materials" in ASM Handbook, 10th ed., Vol. 1, 1990, ASM International, Materials Park, Ohio. ISBN 0871703785.
- ^ Borisenko, Victor E. (2013). Semiconducting Silicides: Basics, Formation, Properties. Springer Science & Business Media. pp. 187, 287. ISBN 978-3-642-59649-0.