Maltotriose
 | |
| Names | |
|---|---|
| IUPAC name
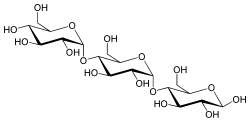
α-D-Glucopyranosyl-(1→4)-α-D-glucopyranosyl-(1→4)-D-glucopyranose
| |
| Systematic IUPAC name
(2R,3S,4S,5S,6R)-2-[(2R,3R,4R,5R,6R)-4,5-dihydroxy-2-(hydroxymethyl)-6-[(2R,3S,4S,5R)-4,5,6-trihydroxy-2-(hydroxymethyl)oxan-3-yl]oxyoxan-3-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.012.886 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C18H32O16 |
| Molar mass | 504.438 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Maltotriose is a trisaccharide (three-part sugar) consisting of three glucose molecules linked with α-1,4 glycosidic bonds.[1]
It is most commonly produced by the digestive enzyme alpha-amylase (a common enzyme in human saliva) on amylose in starch. The creation of both maltotriose and maltose during this process is due to the random manner in which alpha amylase hydrolyses α-1,4 glycosidic bonds.[2]
It is the shortest chain oligosaccharide that can be classified as maltodextrin.
Maltotriose obtained from starch is a common component of ice cream.[3]
References
- ^ Rhoades, Rodney and David R. Bell (2009). Medical Physiology: Principles for Clinical Medicine. Lippincott Williams & Wilkins. ISBN 978-0781768528.
- ^ Schenck, Fred W. (2006). "Glucose and Glucose-Containing Syrups". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a12_457.pub2. ISBN 978-3-527-30385-4.
- ^ Goff, H. Douglas (2015). "Ice Cream and Frozen Desserts". Ullmann's Encyclopedia of Industrial Chemistry. pp. 1–15. doi:10.1002/14356007.a13_563.pub2. ISBN 978-3-527-30673-2.