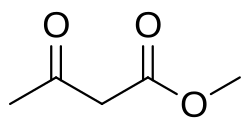
Methyl acetoacetate
 | |
| Names | |
|---|---|
| IUPAC name
methyl 3-oxobutanoate
| |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.003.000 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 1993 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C5H8O3 |
| Molar mass | 116.116 g·mol−1 |
| Appearance | Colourless liquid |
| Odor | Fruit or rum |
| Density | 1.076 g/cm3 |
| Boiling point | 166 °C (331 °F; 439 K) |
Solubility in water
|
40 g/100 mL (20 °C) |
Refractive index (nD)
|
1.411 |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H319 |
Precautionary statements
|
P305+P351+P338 |
| NFPA 704 (fire diamond) | 
2
2 |
| Flash point | 70 °C (158 °F; 343 K) |
| Related compounds | |
Related esters
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
The organic compound methyl acetoacetate is the methyl ester of acetoacetic acid. It is a colorless liquid. It is used as a chemical intermediate. Many of its properties are similar to those for ethyl acetoacetate, which is more common.
At large scale, methyl acetoacetate is industrially produced by treatment of diketene with methanol.[2]
Safety and environmental considerations
Methyl acetoacetate has low toxicity to animals. It is highly biodegradable.[2]
References
- ^ Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 2021-12-19.
- ^ a b Riemenschneider, Wilhelm; Bolt, Hermann M. (2005). "Esters, Organic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a09_565.pub2. ISBN 3527306730.