Methyl dihydrojasmonate
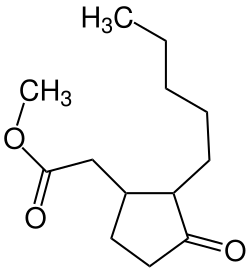 | |
| Names | |
|---|---|
| Preferred IUPAC name
Methyl 2-(3-oxo-2-pentylcyclopentyl)acetate | |
| Other names
Hedione
Kharismal Cepionate | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.042.254 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C13H22O3 |
| Molar mass | 226.316 g·mol−1 |
| Appearance | Clear to pale yellow oily liquid |
| Boiling point | 307.8 °C (586.0 °F; 581.0 K) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Flammable |
| Flash point | 113 °C (235 °F; 386 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Methyl dihydrojasmonate (often referred to by its trade names of hedione or kharismal) is an aroma compound with an odor similar to that of jasmine. It is a synthetic relative of methyl jasmonate, a naturally occurring compound in floral scents such as jasmine, tuberose and magnolia. In racemic mixtures the odor is floral and citrus, while epimerized mixtures exhibit a dense buttery-floral odor with odor recognition thresholds of 15 parts per billion.[1] Considered one of the compounds responsible for the projection of the scent in living flowers, it was first fully characterised and synthesized between 1957 and 1962 in jasmine absolute (0.8%) by the fragrance chemist Edouard Demole, who was working at Firmenich.[2][3][4] and is used in fine fragrances as well as cosmetics, toiletries, and detergents.
Synthesis
Some of the earliest synthesis was based on the selective hydrogenation of methyl jasmonate, which was obtained from natural jasmine oil.[3] However, as this made up <0.8% of the oil, better routes were soon developed. Modern synthesis involves the condensation of cyclopentanone and pentanal, followed by C=C bond isomerisation to give the 2-pentyl-cyclopentenone derivative. Michael reaction of this with dimethyl malonate, followed by decarboxylation gives the desired product.[5]
Use in perfumery
The first commercially successful fragrance to utilise hedione was Eau Sauvage, created by the perfumer Edmond Roudnitska for Christian Dior and launched in 1966. Adding hedione to a classically hesperidic fragrance construction created a dewy lemony magnolia-jasmine dimension without being directly floral. This is considered to be the beginning of a new trend in perfumery towards transparency and projection.[6]
Etymology
The name hedione is derived from Ancient Greek hēdonḗ, "pleasure" (cf. hedonism). Kharismal is derived from the Greek kharisma, meaning "grace" or "favor" (cf. charisma).
References
- ^ Leffingwell, John C. (2001), The Methyl dihydrojasmonates, Leffingwell & Associates
- ^ Theimer, Ernst T (2014). Fragrance Chemistry Science of the Sense of Smell, The. Elsevier Science. ISBN 978-0-323-13860-4. OCLC 1044713310.
- ^ a b Demole, E.; Lederer, E.; Mercier, D. (January 1962). "Synthèse du dihydrojasmonate de méthyle (pentyl-2-oxo-3-cyclopentylacétate de méthyle)". Helvetica Chimica Acta. 45 (2): 685–692. doi:10.1002/hlca.19620450234.
- ^ Demole, E.; Lederer, E.; Mercier, D. (January 1962). "Isolement et détermination de la structure du jasmonate de méthyle, constituant odorant caractéristique de l'essence de jasmin". Helvetica Chimica Acta. 45 (2): 675–685. doi:10.1002/hlca.19620450233.
- ^ Chapuis, Christian (September 2012). "The Jubilee of Methyl Jasmonate and Hedione ®". Helvetica Chimica Acta. 95 (9): 1479–1511. doi:10.1002/hlca.201200070.
- ^ Berger, Ralf Günter (2012). "Scent and Chemistry. The Molecular World of Odors. By Günther Ohloff, Wilhelm Pickenhagen and Philip Kraft". Angewandte Chemie International Edition. 51 (13). Verlag GmbH & Co. KGaA, Weinheim: 3058. doi:10.1002/anie.201201256.