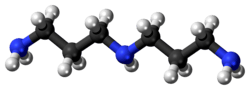
Norspermidine
 | |
| Names | |
|---|---|
| Preferred IUPAC name
N1-(3-Aminopropyl)propane-1,3-diamine | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
1071254 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.238 |
| EC Number |
|
Gmelin Reference
|
26839 |
| KEGG | |
| MeSH | norspermidine |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2269 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H17N3 |
| Molar mass | 131.223 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Ichtyal, ammoniacal |
| Density | 938 mg mL−1 |
| Melting point | −16 to 0 °C; 3 to 32 °F; 257 to 273 K |
| Boiling point | 240.60 °C; 465.08 °F; 513.75 K |
| log P | −0.826 |
Refractive index (nD)
|
1.481–1.482 |
| Hazards | |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H302, H311, H314, H317, H330 |
Precautionary statements
|
P260, P280, P284, P305+P351+P338, P310 |
| Flash point | 117 °C (243 °F; 390 K) |
Autoignition
temperature |
280 °C (536 °F; 553 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
738 mg kg−1 (oral, rat) |
| Safety data sheet (SDS) | fishersci.com |
| Related compounds | |
Related amines
|
|
Related compounds
|
Agmatine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Norspermidine is a polyamine of similar structure to the more common spermidine. Norspermidine has been found to occur naturally in some species of plants,[1][2] bacteria,[3] and algae.[4]
Norspermidine is being researched for use as a cancer medication.[5][6]
Biosynthesis
Norspermidine is an aliphatic polyamine. In ϵ-proteobacteria, which are found in human gut microbiota, a combination of two enzymes is used to produce norspermidine from 1,3-diaminopropane.[7] First, carboxynorspermidine synthase catalyses a reductive amination using nicotinamide adenine dinucleotide phosphate (NADPH) as the reducing agent.[8][9]
The intermediate, carboxynorspermidine, is then decarboxylated by carboxynorspermidine decarboxylase:[7][9]
References
- ^ Rodriguez-Garay, B; et al. (1989). "Detection of Norspermidine and Norspermine in Medicago sativa L. (Alfalfa)". Plant Physiology. 89 (2): 525–529. doi:10.1104/pp.89.2.525. ISSN 0032-0889. PMC 1055875. PMID 16666576.
- ^ Hamana, K; et al. (1998). "Unusual polyamines in aquatic plants: the occurrence of homospermidine, norspermidine, thermospermine, norspermine, aminopropylhomospermidine, bis(aminopropyl)ethanediamine, and methylspermidine". Can. J. Bot. 76 (1): 130–133. doi:10.1139/cjb-76-1-130.
- ^ Yamamoto, S; et al. (Apr 27, 1979). "Occurrence of norspermidine in some species of genera Vibrio and Beneckea". Biochem Biophys Res Commun. 87 (4): 1102–1108. doi:10.1016/S0006-291X(79)80021-2. PMID 313792.
- ^ Hamana, K; Matsuzaki, S (1982). "Widespread Occurrence of Norspermidine and Norspermine in Eukaryotic Algae". J. Biochem. 91 (4): 1321–1328. doi:10.1093/oxfordjournals.jbchem.a133818. ISSN 0021-924X. PMID 7096289.
- ^ Prakash, NJ; et al. (1988). "Antitumor activity of norspermidine, a structural homologue of the natural polyamine spermidine". Anticancer Res. 8 (4): 563–568. PMID 3140710.
- ^ Sunkara, PS; et al. (1988). "Mechanism of antitumor activity of norspermidine, a structural homologue of spermidine". Progress in Polyamine Research. Adv Exp Med Biol. Vol. 250. pp. 707–716. doi:10.1007/978-1-4684-5637-0_62. ISBN 978-1-4684-5639-4. PMID 3255245.
- ^ a b Hanfrey CC, Pearson BM, Hazeldine S, Lee J, Gaskin DJ, Woster PM, Phillips MA, Michael AJ (December 2011). "Alternative spermidine biosynthetic route is critical for growth of Campylobacter jejuni and is the dominant polyamine pathway in human gut microbiota". The Journal of Biological Chemistry. 286 (50): 43301–12. doi:10.1074/jbc.M111.307835. PMC 3234850. PMID 22025614.
- ^ Nakao H, Shinoda S, Yamamoto S (July 1991). "Purification and some properties of carboxynorspermidine synthase participating in a novel biosynthetic pathway for norspermidine in Vibrio alginolyticus". Journal of General Microbiology. 137 (7): 1737–42. doi:10.1099/00221287-137-7-1737. PMID 1955861.
- ^ a b Lee J, Sperandio V, Frantz DE, Longgood J, Camilli A, Phillips MA, Michael AJ (April 2009). "An alternative polyamine biosynthetic pathway is widespread in bacteria and essential for biofilm formation in Vibrio cholerae". The Journal of Biological Chemistry. 284 (15): 9899–907. doi:10.1074/jbc.M900110200. PMC 2665113. PMID 19196710.
amin_Struktur.svg.png)

-2-Amino-4-oxobutanoic_acid.svg.png)