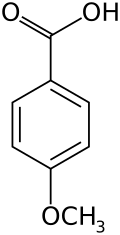
p-Anisic acid
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
4-Methoxybenzoic acid | |||
| Other names
Draconic acid
| |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
|||
Beilstein Reference
|
508910 | ||
| ChEBI | |||
| ChEMBL |
| ||
| ChemSpider | |||
| ECHA InfoCard | 100.002.562 | ||
| EC Number |
| ||
Gmelin Reference
|
3499 | ||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
C8H8O3 | ||
| Molar mass | 152.149 g·mol−1 | ||
| Density | 1.385 g/cm3 | ||
| Melting point | 184 °C (363 °F; 457 K) (sublimation) | ||
| Boiling point | 275 to 280 °C (527 to 536 °F; 548 to 553 K) | ||
Solubility in water
|
1 part per 2500 | ||
| Acidity (pKa) | 4.47[2] | ||
| Structure[3] | |||
Crystal structure
|
monoclinic | ||
Space group
|
P21/a | ||
Lattice constant
|
a = 16.98 Å, b = 10.95 Å, c = 3.98 Å α = 90°, β = 98.7°, γ = 90°
| ||
Formula units (Z)
|
4 | ||
| Hazards | |||
| GHS labelling:[4] | |||
Pictograms
|

| ||
Signal word
|
Warning | ||
Hazard statements
|
H302, H315, H319, H335 | ||
Precautionary statements
|
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P330, P332+P313, P337+P313, P362, P403+P233, P405 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
p-Anisic acid, also known as 4-methoxybenzoic acid or draconic acid, is one of the isomers of anisic acid. The term "anisic acid" often refers to this form specifically.[1] It is a white crystalline solid which is insoluble in water, highly soluble in alcohols, and soluble in ether and ethyl acetate.[1]
Synthesis and occurrence
p-Anisic acid is found naturally in anise. It was first synthesized in 1841 by Auguste Cahours by oxidizing anethole that he had isolated from anise by recrystallization with diluted nitric acid:[5][6]
- CH3CH=CHC6H4OCH3 + HNO3 → CH3OC6H4CHO + others
- CH3OC6H4CHO + HNO3 → CH3OC6H4COOH + others
Oxidation of anisaldehyde, which was Cahours' intermediate product, is still used nowadays.[7] Anisic acid can also be obtained synthetically by the oxidation of p-methoxyacetophenone.
Uses
p-Anisic acid has antiseptic properties.[8] It is also used as an intermediate in the preparation of more complex organic compounds.
References
- ^ a b c Merck Index, 11th Edition, 696
- ^ Braude, E. A.; Nachod, F. C., eds. (1955). Determination of Organic Structure by Physical Methods. Academic Press. ISBN 9781483275727.
{{cite book}}: ISBN / Date incompatibility (help) - ^ Bryan, Robert F. (1967). "An X-ray study of the p-n-alkoxybenzoic acids. Part II. The crystal structure of anisic acid". Journal of the Chemical Society B: Physical Organic: 1311–1316. doi:10.1039/j29670001311. ISSN 0045-6470.
- ^ "4-Methoxybenzoic acid". pubchem.ncbi.nlm.nih.gov. Retrieved 22 December 2021.
- ^ Wisniak, Jaime (2013-10-01). "Auguste André Thomas Cahours". Educación Química. 24 (4): 451–460. doi:10.1016/S0187-893X(13)72500-X. ISSN 0187-893X.
- ^ Crochard (París); Arago, François; Gay-Lussac, Joseph Louis (1841). Annales de chimie et de physique (in French). Chez Crochard.
- ^ Saha, Rumpa; Ghosh, Aniruddha; Saha, Bidyut (2013). "Kinetics of micellar catalysis on oxidation of p-anisaldehyde to p-anisic acid in aqueous medium at room temperature". Chemical Engineering Science. 99: 23–27. Bibcode:2013ChEnS..99...23S. doi:10.1016/j.ces.2013.05.043.
- ^ Herman, Anna (2019). "Antimicrobial Ingredients as Preservative Booster and Components of Self-Preserving Cosmetic Products". Current Microbiology. 76 (6): 744–754. doi:10.1007/s00284-018-1492-2. PMID 29651551.