Phenyl isothiocyanate
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Isothiocyanatobenzene[1] | |||
| Other names
Phenyl isothiocyanate[1]
Thiocarbanil | |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.002.853 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
C7H5NS | ||
| Molar mass | 135.19 g/mol | ||
| Appearance | Colorless liquid with a pungent odor[2] | ||
| Density | 1.1288 g/cm3[2] | ||
| Melting point | −21 °C (−6 °F; 252 K)[3] | ||
| Boiling point | 221 °C (430 °F; 494 K)[3] | ||
Solubility in water
|
negligible [2] | ||
| Solubility | ethanol, ether[3] | ||
Magnetic susceptibility (χ)
|
−86.0·10−6 cm3/mol | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
toxic, flammable[2] | ||
| GHS labelling: | |||
Pictograms
|
   [3] [3]
| ||
Signal word
|
Danger[3] | ||
Hazard statements
|
H301, H311, H314, H317, H331, H334, H361[3] | ||
Precautionary statements
|
P261, P280, P301+P310, P301+P330+P331, P302+P350, P304+P341, P305+P351+P338, P310, P312, P342+P311[3] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Phenyl isothiocyanate (PITC) is a reagent for organic chemistry. Also known as Edman's reagent, it is used in Edman degradation.
Edman degradation's main competitor uses ortho-phthaldehyde (OPA), which is less sensitive and thus amenable to automated, reversed-phase HPLC. However, PITC can be used for analysing secondary amines, unlike OPA.
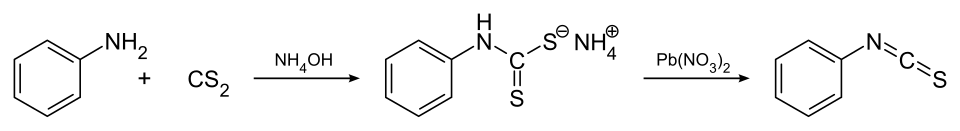
Commercially available, this compound may be synthesized by the reaction of aniline with carbon disulfide and concentrated ammonia to give the ammonium dithiocarbamate salt of aniline in the first step, which on further reaction with lead(II) nitrate gives phenyl isothiocyanate:[4]
Another method of synthesizing this reagent involves a Sandmeyer reaction using aniline, sodium nitrite and copper(I) thiocyanate.
See also
- Isothiocyanate
- Naphthyl isothiocyanate
References
- ^ a b Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 665. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ a b c d "Phenyl isothiocyanate - CAS # 103-72-0".
- ^ a b c d e f g "Message".
- ^ F. B. Dains, R. Q. Brewster, and C. P. Olander (1926). "Phenyl isothiocyanate". Organic Syntheses. 6: 72. doi:10.15227/orgsyn.006.0072
{{cite journal}}: CS1 maint: multiple names: authors list (link).