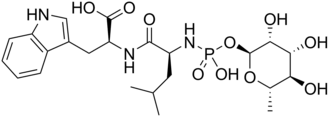
Phosphoramidon
 | |
 | |
| Names | |
|---|---|
| IUPAC name
(2S)-2-[[(2S)-2-[[hydroxy-[(2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxyphosphoryl]amino]-4-methylpentanoyl]amino]-3-(1H-indol-3-yl)propanoic acid
| |
| Other names
N-[N-[[(6-deoxy-α-L-mannoopyranosyl)oxy]hydroxyphosphinyl]-L-leucyl]-L-tryptophan
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.048.164 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C23H34N3O10P |
| Molar mass | 543.510 g·mol−1 |
| Appearance | White to slightly yellow solid |
Solubility in water
|
Soluble as sodium salt[1] |
| Solubility in DMSO and methanol | Soluble as sodium salt[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Phosphoramidon is a chemical compound derived from cultures of Streptomyces tanashiensis. It is an inhibitor of the enzyme thermolysin,[2] of the membrane metallo-endopeptidase,[3] and of the endothelin converting enzyme.[3][4] Chemically, phosphoramidon differs from its closely related peptidase inhibitor talopeptin by a single stereocenter.
Because of its enzyme inhibitory properties, phosphoramidon is widely used as a biochemical tool.
References
- ^ a b Phosphoramidon, Enzo Life Sciences
- ^ Kitagishi K, Hiromi K (1984). "Binding between thermolysin and its specific inhibitor, phosphoramidon". Journal of Biochemistry. 95 (2): 529–34. doi:10.1093/oxfordjournals.jbchem.a134635. PMID 6715312.
- ^ a b Phosphoramidon at PubChem
- ^ Schulz, Henk; Dale, Glenn E.; Karimi-Nejad, Yasmin; Oefner, Christian (2009-01-09). "Structure of human Endothelin-converting Enzyme I Complexed with Phosphoramidon". Journal of Molecular Biology. 385 (1): 178–187. doi:10.1016/j.jmb.2008.10.052. ISSN 0022-2836.
External links
- The MEROPS online database for peptidases and their inhibitors: Phosphoramidon