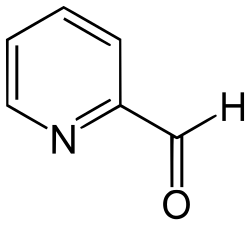
Pyridine-2-carbaldehyde
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Pyridine-2-carbaldehyde | |
| Other names
Picolinaldehyde
2-Pyridine carboxaldehyde 2-Pyridyl aldehyde Picolinal | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.013.031 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H5NO |
| Molar mass | 107.112 g·mol−1 |
| Density | 1.126 g/mL |
| Melting point | 148–151 °C (298–304 °F; 421–424 K) |
| Boiling point | 181 °C (358 °F; 454 K) |
| Related compounds | |
Related Aldehydes
|
Salicylaldehyde Quinoline Carboxaldehyde |
Related compounds
|
Picolinic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Pyridine-2-carbaldehyde, also called 2-formylpyridine, is an organic compound with the formula NC5H4CHO. It is one of three isomeric pyridinaldehydes. The other isomers are pyridine-3-carboxaldehyde and pyridine-4-carboxaldehyde.
Pyridine-2-carbaldehyde is a colorless oily liquid with a distinctive odor. Older samples are often brown-colored owing to impurities. It serves as a precursor to other compounds of interest in coordination chemistry and pharmaceuticals. Pyridine aldehydes are typically prepared by oxidation of the hydroxymethyl- or methylpyridines.[1]
Reactions and uses
The drug pralidoxime can be produced from 2-formylpyridine.
The aldehyde functional group is subject to nucleophilic attack, specifically by amines to form Schiff bases, which serve as bidentate ligands.[2] Iminopyridine complexes can be remarkably robust.[3]
Pyridine-2-carbaldehyde is used in the synthesis of ARN2966 [102212-26-0],[4] one of the syntheses of Mefloquine,[5] RWJ-22757 (Mcn-5195) [103729-18-6],[6] & in one of the syntheses of Carbinoxamine.[7][8]
References
- ^ Shimizu, Shinkichi; Watanabe, Nanao; Kataoka, Toshiaki; Shoji, Takayuki; Abe, Nobuyuki; Morishita, Sinji; Ichimura, Hisao (2007). "Pyridine and Pyridine Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a22_399. ISBN 978-3-527-30673-2.
- ^ Chatterjeea, D.; Mitra, A. (2004). "Synthesis, Characterization and Reactivities of Schiff-base Complexes of Ruthenium(III)". J. Coord. Chem. 57: 175–182. doi:10.1080/00958970410001662435.
- ^ Mal, P.; Breiner, B.; Rissanen, K.; Nitschke, J. R. (2009). "White Phosphorus is Air-Stable Within a Self-Assembled Tetrahedral Capsule". Science. 324 (5935): 1697–1699. doi:10.1126/science.1175313. PMID 19556504.
- ^ Bijan Almassian, Martin J. Sadowski, & Xu Kevin Lin, US8658677 (2014 to New York University NYU, Aria Neurosciences Inc).
- ^ Guenther North Caldwell N.J. Us Grethe, Toomas Bloomfield N.J. Us Mitt, DE2806909 (1988 to F. Hoffmann-La Roche & Co. Ag, Basel, Ch).
- ^ Richard J. Carmosin & John R. Carson, US4582836 (1986 to Janssen Pharmaceuticals Inc).
- ^ A. P. Swain, US2800485 (1957).
- ^ 王启发, 程青芳, 刘洲, CN104774174 (2017 to 淮海工学院).