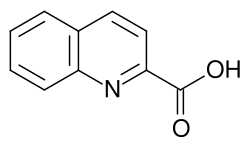
Quinaldic acid
 | |
| Names | |
|---|---|
| IUPAC name
quinoline-2-carboxylic acid
| |
| Other names
2-Carboxyquinoline
2-Quinolinecarboxylic acid | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
126322 |
| ChEBI | |
| ChEMBL | |
| DrugBank | |
| ECHA InfoCard | 100.002.018 |
| EC Number |
|
Gmelin Reference
|
143145 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C10H7NO2 |
| Molar mass | 173.171 g·mol−1 |
Solubility in water
|
14000 mg/L |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319, H335 |
Precautionary statements
|
P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Quinaldic acid is an organic chemical compound.[1][2][3][4]
Quinaldic acid can form a zwitterion, where a proton is transferred from the carboxylic acid group to the nitrogen atom.[5] Neptunium and uranium can form mixed ligand compounds with quinaldic acid.[5]
Quinaldic acid has been used in the gravimetric analysis of uranium in the form of uranyl ion. This is precipitated by forming an insoluble salt with quinaldic acid. When incinerated U3O8 is formed and can be weighed.[6]
References
- ^ PubChem. "Quinaldic Acid". pubchem.ncbi.nlm.nih.gov. Retrieved 2025-12-09.
- ^ "Quinaldic Acid". go.drugbank.com. Retrieved 2025-12-09.
- ^ PubChem. "Quinaldic acid, 1,2,3,4-tetrahydro-8-hydroxy-4-oxo-, L-". pubchem.ncbi.nlm.nih.gov. Retrieved 2025-12-09.
- ^ Tajieva, Galiya R.; Ali Siddique, Sabir; Ibragimov, Aziz B.; Mohamed, Shaaban K.; Ibragimova, Mavlyuda R.; Abuelizz, Hatem A.; Al-Salahi, Rashad; M.Ashurov, Jamshid; Ibragimov, Bakhtiyar T.; Gao, Junkuo; El Bakri, Youness (2024-08-01). "Synthesis and characterization of novel quinaldic acid based Ni, Fe, and Cr Complexes: A computational and experimental study". Inorganic Chemistry Communications. 166 112590. doi:10.1016/j.inoche.2024.112590. ISSN 1387-7003.
- ^ a b Andreev, Grigory; Budantseva, Nina; Fedoseev, Aleksander (September 2022). "The novel U(VI) and Np(VI) mixed-ligand complexes based on quinaldic acid zwitterion". Polyhedron. 224 116003. doi:10.1016/j.poly.2022.116003.
- ^ Ware, Elinor (1947). Organic Reagents for Uranium Analysis. Technical Information Division, Oak Ridge Operations. p. 18.