Remikiren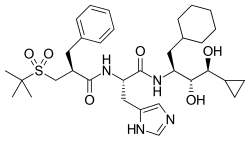 |
|
| ATC code | |
|---|
|
| Legal status |
|
|---|
|
(2S)-2-[(2R)-2-benzyl-3-(2-methylpropane-2-sulfonyl)propanamido]-N-[(2R,3S,4R)-1-cyclohexyl-4-cyclopropyl-3,4-dihydroxybutan-2-yl]-3-(1H-imidazol-4-yl)propanamide
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
|
| Formula | C33H50N4O6S |
|---|
| Molar mass | 630.85 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
O=S(=O)(C(C)(C)C)C[C@H](C(=O)N[C@H](C(=O)N[C@@H](CC1CCCCC1)[C@@H](O)[C@@H](O)C2CC2)Cc3cnc[nH]3)Cc4ccccc4
|
InChI=1S/C33H50N4O6S/c1-33(2,3)44(42,43)20-25(16-22-10-6-4-7-11-22)31(40)37-28(18-26-19-34-21-35-26)32(41)36-27(17-23-12-8-5-9-13-23)30(39)29(38)24-14-15-24/h4,6-7,10-11,19,21,23-25,27-30,38-39H,5,8-9,12-18,20H2,1-3H3,(H,34,35)(H,36,41)(H,37,40)/t25-,27+,28+,29+,30-/m1/s1  Y YKey:UXIGZRQVLGFTOU-VQXQMPIVSA-N  Y Y
|
| (verify) |
Remikiren is a renin inhibitor under development for the treatment of hypertension (high blood pressure). It was first developed by Hoffmann–La Roche in 1996.[1]
References
- ^ Richter WF, Whitby BR, Chou RC (1996). "Distribution of remikiren, a potent orally active inhibitor of human renin, in laboratory animals". Xenobiotica. 26 (3): 243–54. doi:10.3109/00498259609046705. PMID 8730917.
Antihypertensive drugs acting on the renin–angiotensin system (C09) |
|---|
ACE inhibitors
("-pril") |
- Sulfhydryl-containing: Captopril
- Rentiapril
- Zofenopril (+nebivolol)
- Dicarboxylate-containing: Enalapril# (+lercanidipine, +nitrendipine)
- Benazepril (+amlodipine, +pimobendan)
- Cilazapril
- Delapril (+manidipine)
- Imidapril
- Lisinopril (+amlodipine, +HCT)
- Moexipril
- Perindopril (+amlodipine, +bisoprolol, +indapamide, +amlodipine and indapamide, +bisoprolol and amlodipine, +bisoprolol, amlodipine, and indapamide)
- Quinapril (+HCT)
- Ramipril (+amlodipine, +amlodipine and HCT, +bisoprolol, +felodipine)
- Spirapril
- Temocapril
- Trandolapril (+verapamil)
- Phosphonate-containing: Ceronapril
- Fosinopril (+HCT)
- Other/ungrouped: Alacepril
|
|---|
AIIRAs
("-sartan") |
- Azilsartan
- Candesartan (+amlodipine, +amlodipine and HCT)
- Eprosartan
- Fimasartan
- Irbesartan (+amlodipine, +amlodipine and HCT, +HCT)
- Losartan (+amlodipine, +HCT)
- Olmesartan (+amlodipine, +amlodipine and HCT, +HCT)
- Tasosartan§
- Telmisartan (+amlodipine, +amlodipine and HCT, +amlodipine and indapamide, +HCT)
- Valsartan (+aliskiren, +amlodipine, +amlodipine and HCT, +HCT, +lercanidipine, +nebivolol, +sacubitril)
|
|---|
Renin inhibitors
("-kiren") | |
|---|
| Dual ACE/NEP inhibitors |
- Gemopatrilat
- Ilepatril
- Omapatrilat
- Sampatrilat
|
|---|
| Neprilysin inhibitors | |
|---|
| Other | |
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
Angiotensin receptor modulators |
|---|
| ATRTooltip Angiotensin receptor |
- Agonists: Angiotensin II
- Angiotensin III
- Angiotensin IV
- L-163,491
- Saralasin
- Propeptides: Angiotensinogen
- Angiotensin I
|
|---|
| Combinations: |
- Amlodipine/valsartan
- Olmesartan/amlodipine
- Olmesartan/amlodipine/hydrochlorothiazide
- Valsartan/hydrochlorothiazide
- Valsartan/hydrochlorothiazide/amlodipine
|
|---|
