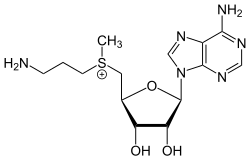
S-Adenosylmethioninamine
 | |
| Names | |
|---|---|
| IUPAC name
S-(3-Aminopropyl)-S-methyl-5′-thioadenosin-5′-ium
| |
| Systematic IUPAC name
(3-Aminopropyl){[(2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methyl}methylsulfanium | |
| Other names
S-Adenosyl-(5′)-3-methylthiopropylamine
decarboxylated S-adenosyl methionine decarboxy-S-adenosyl methionine (5-Deoxy-5-adenosyl)(3-aminopropyl)methylsulfonium cation | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| Abbreviations | dAdoMet, dc-SAM |
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C14H23N6O3S+ |
| Molar mass | 355.43582 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
S-Adenosylmethioninamine is a substrate that is required for the biosynthesis of polyamines including spermidine, spermine, and thermospermine.[1] It is produced by decarboxylation of S-adenosyl methionine.
This reaction is catalyzed by S-adenosylmethionine decarboxylase.[2] The enzyme binds to S-adenosyl methionine (SAM) and removes the carboxyl group from the methionine. Once formed, S-adenosylmethioninamine donates its aminopropyl group to synthesize polyamines[3]. Polyamines are important for DNA stability, RNA function, and cell growth.[4]
See also
- Adenosylmethionine decarboxylase (AMD1)
- Spermidine synthase
- Spermine synthase
- Thermospermine synthase (ACAULIS5)
References
- ^ Takahashi, Taku; Kakehi, Jun-Ichi (2009-10-13). "Polyamines: Ubiquitous polycations with unique roles in growth and stress responses". Annals of Botany. 105 (1): 1–6. doi:10.1093/aob/mcp259. PMC 2794062. PMID 19828463.
- ^ Campbell, Mary K.; Farrell, Shawn O.; McDougal, Owen M. (2016). Biochemistry (9th ed.). Boston, MA: Cengage Learning. ISBN 978-1-305-96113-5.
- ^ Sirasunthorn, Nichanun; Jailwala, Anuj; Gerber, Anna; Comstock, Lindsay R. (2019-09-18). "Evaluation of N ‐Mustard Analogues of S ‐Adenosyl‐L‐methionine with Eukaryotic DNA Methyltransferase 1". ChemistrySelect. 4 (35): 10525–10531. doi:10.1002/slct.201902940. ISSN 2365-6549.
- ^ Takahashi, Taku; Kakehi, Jun-Ichi (2009-10-13). "Polyamines: Ubiquitous polycations with unique roles in growth and stress responses". Annals of Botany. 105 (1): 1–6. doi:10.1093/aob/mcp259. PMC 2794062. PMID 19828463.