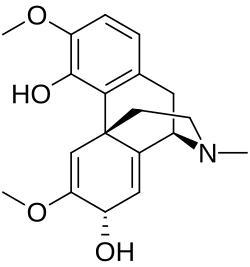
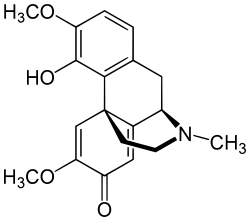
Salutaridine
 | |
 | |
| Names | |
|---|---|
| IUPAC name
4-Hydroxy-3,6-dimethoxy-17-methyl-5,6,8,14-tetradehydromorphinan-7-one
| |
| Systematic IUPAC name
(4aS,10R)-5-Hydroxy-3,6-dimethoxy-11-methyl-9,10-dihydro-2H-10,4a-(azanoethano)phenanthren-2-one | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C19H21NO4 |
| Molar mass | 327.380 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Salutaridine, also known as floripavine, is an alkaloid that is present in the morphinan alkaloid pathway of opium poppy, Papaver somniferum.
Biosynthesis
It is produced by the enzyme salutaridine synthase that catalyzes the chemical reaction from its precursor (R)-reticuline:[1][2]
The enzyme uses reduced nicotinamide adenine dinucleotide phosphate (NADPH) as its cofactor.
Salutaridine is converted to salutaridinol by the enzyme salutaridine reductase (NADPH).[3]
References
- ^ Enzyme 1.14.19.67 at KEGG Pathway Database.
- ^ Gerady R, Zenk MH (1993). "Formation of salutaridine from (R)-reticuline by a membrane-bound cytochrome P-450 enzyme from Papaver somniferum". Phytochemistry. 32: 79–86. doi:10.1016/0031-9422(92)80111-Q.
- ^ Gerady R, Zenk MH (1993). "Purification and characterization of salutaridine:NADPH 7-oxidoreductase from Papaver somniferum". Phytochemistry. 34 (1): 125–132. Bibcode:1993PChem..34..125G. doi:10.1016/S0031-9422(00)90793-3.
-reticuline.svg.png)