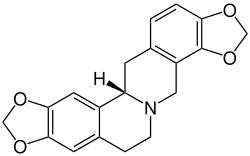
Tetrahydrocoptisine
 (S)-tetrahydrocoptisine
| |
| Names | |
|---|---|
| IUPAC name
5,7,17,19-tetraoxa-13-azahexacyclo[11.11.0.02,10.04,8.015,23.016,20]tetracosa-2,4(8),9,15(23),16(20),21-hexaene
| |
| Other names
Stylopine
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C19H17NO4 |
| Molar mass | 323.348 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Tetrahydrocoptisine (also known as stylopine) is an alkaloid isolated from Corydalis species.[1][2]
Biosynthesis
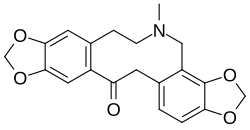
The (S)-isomer of tetrahydrocoptisine is produced when the enzyme (S)-stylopine synthase acts on (S)-cheilanthifoline to form a second methylenedioxy ring:[2][3]
Metabolism
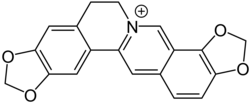
Tetrahydrocoptisine is converted to coptisine by an oxidation reaction catalysed by the enzyme tetrahydroberberine oxidase.[2][4]

Alternatively, it can be converted into protopine in two steps. The first is a methylation reaction by the enzyme (S)-tetrahydroprotoberberine N-methyltransferase using the cofactor, S-adenosyl methionine (SAM). This transfers a methyl group, giving S-adenosyl-L-homocysteine (SAH).[2][5]
Then the product, (S)-cis-N-methylstylopine, is oxidised by the enzyme methyltetrahydroprotoberberine 14-monooxygenase:[2][6]
References
- ^ Li, W.; Huang, H.; Zhang, Y.; Fan, T.; Liu, X.; Xing, W.; Niu, X. (2013). "Anti-inflammatory effect of tetrahydrocoptisine from Corydalis impatiens is a function of possible inhibition of TNF-α, IL-6 and NO production in lipopolysaccharide-stimulated peritoneal macrophages through inhibiting NF-κB activation and MAPK pathway". European Journal of Pharmacology. 715 (1–3): 62–71. doi:10.1016/j.ejphar.2013.06.017. PMID 23810685.
- ^ a b c d e Tian, Ya; Kong, Lingzhe; Li, Qi; Wang, Yifan; Wang, Yongmiao; An, Zhoujie; Ma, Yuwei; Tian, Lixia; Duan, Baozhong; Sun, Wei; Gao, Ranran; Chen, Shilin; Xu, Zhichao (2024). "Structural diversity, evolutionary origin, and metabolic engineering of plant specialized benzylisoquinoline alkaloids". Natural Product Reports. 41 (11): 1787–1810. doi:10.1039/D4NP00029C. PMID 39360417.
- ^ Bauer W, Zenk MH (1991). "Two methylenedioxy bridge-forming cytochrome P-450 dependent enzymes are involved in (S)-stylopine biosynthesis". Phytochemistry. 30 (9): 2953–2961. Bibcode:1991PChem..30.2953B. doi:10.1016/S0031-9422(00)98230-X.
- ^ "Coptisine biosynthesis". PubChem. Retrieved 2026-02-06.
- ^ Rueffer M, Zumstein G, Zenk MH (1990). "Partial purification and characterization of S-adenosyl-L-methionine:(S)-tetrahydroprotoberberine cis-N-methyltransferase from suspension-cultured cells of Eschscholtzia and Corydalis" (PDF). Phytochemistry. 29 (12): 3727–3733. doi:10.1016/0031-9422(90)85321-6.
- ^ Rueffer M, Zenk MH (1987). "Enzymatic formation of protopines by a microsomal cytochrome-P-450 system of Corydalis vaginans". Tetrahedron Lett. 28 (44): 5307–5310. doi:10.1016/S0040-4039(00)96715-7.
-(S)-Cheilanthifoline.svg.png)
-cis-N-methylstylopine.svg.png)