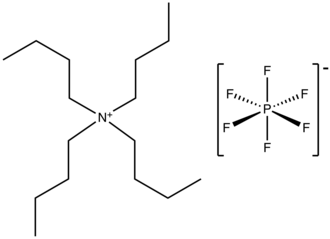
Tetrabutylammonium hexafluorophosphate
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Tetrabutylammonium hexafluorophosphate
| |
| Other names
1-Butanaminium, N,N,N-tributyl-, hexafluorophosphate(1-)
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.019.520 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C16H36F6NP |
| Molar mass | 387.4279 g·mol−1 |
| Appearance | white powder |
| Melting point | 244–246 °C (471–475 °F; 517–519 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Tetrabutylammonium hexafluorophosphate is a salt with the formula NBu4PF6. It is a white powder that is used as an electrolyte in nonaqueous electrochemistry. It is highly soluble in polar organic solvents such as acetone and acetonitrile.
The salt consists of a positively charged tetrabutylammonium, a quaternary ammonia cation and a weakly basic hexafluorophosphate anion. These species are chemically inert, which allows the salt to serve as an inert electrolyte over a wide potential range.
Preparation
This salt can be prepared by the reaction of tetrabutylammonium bromide and potassium hexafluorophosphate in aqueous solution.[1]
- (C4H9)4NBr + KPF6 → (C4H9)4NPF6 + KBr
Given the sensitivity of electrochemical experiments, this salt is usually further purified, e.g., by recrystallization from aqueous or absolute ethanol.[2]
Use
This salt is used as a supporting electrolyte in electroanalysis as well as electrolytic growth methods (electrocrystallization). For instance, in the presence of this salt, the electrochemical oxidation of tetramethyltetraselenafulvalene (TMTSF) yields single crystals of (TMTSF)2PF6. This material is the first organic superconductor (Tc = 0.9 K at 12 kbar) reported by Klaus Bechgaard and Denis Jérome in 1980.[3]
References
- ^ Eliassaf, Jehudah; Fuoss, Raymond M.; Lind Jr., John E. (1963). "Conductance of Quaternary Ammonium Hexafluorophosphates in acetonitrile". The Journal of Physical Chemistry. 67 (9): 1941–1942. doi:10.1021/j100803a519.
- ^ Zoski, Cynthia G. (2007). Handbook of Electrochemistry (1st ed.). Amsterdam: Elsevier.
- ^ Jérome, D.; Mazaud, A.; Ribault, M.; Bechgaard, K. (1980). "Superconductivity in a synthetic organic conductor (TMTSF)2PF6". Journal de Physique Lettres. 41: 95–98. doi:10.1051/jphyslet:0198000410409500.