Tetraethylsilane
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Tetraethylsilane[1]
| |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.010.142 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Si(CH2CH3)4 |
| Molar mass | 144.333 g·mol−1 |
| Appearance | Colorless liquid[2] |
| Odor | Distinct, mild[3] |
| Density | 0.761 g/cm3[2] |
| Melting point | −82.5 °C (−116.5 °F; 190.7 K)[2] |
| Boiling point | 153–154 °C (307–309 °F; 426–427 K)[2] |
Solubility in water
|
Almost insoluble in water[4] |
| Vapor pressure | 5 mmHg at 20 °C (68 °F)[3] |
Refractive index (nD)
|
1.426[2] |
| Structure | |
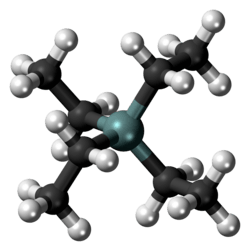
Molecular shape
|
Tetrahedral at Si |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Flammable |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Warning |
Hazard statements
|
H226, H315, H319, H335 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P261, P264, P264+P265, P271, P280, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P370+P378, P403+P233, P403+P235, P405, P501 |
| Flash point | 25 °C[2] |
Autoignition
temperature |
235 °C (455 °F; 508 K)[3] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Tetraethylsilane is an organosilicon compound with the chemical formula Si(CH2CH3)4. It is a colorless liquid. Under standard coditions, it does not react with water, air, nitric acid or concentrated potassium hydroxide.[4] Its vapor density is 4.98, relative to air (air = 1).[2]
Synthesis
The synthesis of tetraethylsilane by reacting silicon tetrachloride with diethylmercury or diethylzinc in an ampoule at 140–160 °C (284–320 °F) was first reported in 1863 by Charles Friedel and James Mason Crafts.[4][5]
- SiCl4 + 2 Zn(CH2CH3)2 → Si(CH2CH3)4 + 2 ZnCl2
Uses
Tetraethylsilane reacts with halides of the boron group elements, such as boron tribromide or gallium trichloride with high yield and purity to form the corresponding dihaloethyl compound.[6]
- Si(CH2CH3)4 + BBr3 → (CH3CH2)3SiBr + CH3CH2BBr2
- Si(CH2CH3)4 + GaCl3 → (CH3CH2)3SiCl + CH3CH2GaCl2
References
- ^ a b PubChem. "Tetraethylsilane". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-02-11.
- ^ a b c d e f g "Tetraethylsilane". sigmaaldrich.com. Retrieved February 11, 2026.
{{cite web}}: CS1 maint: url-status (link) - ^ a b c "TETRAETHYLSILANE" (PDF). gelest.com. October 23, 2014. Retrieved February 11, 2026.
{{cite web}}: CS1 maint: url-status (link) - ^ a b c Friedel, C.; Crafts, J. M. (January 1863). "Ueber einige neue organische Verbindungen des Siliciums und das Atomgewicht dieses Elementes". Justus Liebigs Annalen der Chemie. 127 (1): 28–32. doi:10.1002/jlac.18631270103. ISSN 0075-4617.
- ^ Soukup, Rudolf Werner (2020). Chemiegeschichtliche Daten organischer Substanzen [Chemical history data of organic substances] (in German).
- ^ Schmidbaur, H.; Findeiss, W. (October 1964). "A Simple Route to Organogallium Compounds". Angewandte Chemie International Edition in English. 3 (10): 696–696. doi:10.1002/anie.196406961. ISSN 0570-0833. Archived from the original on 2021-07-22.